Vardaan Learning Institute
Class 11 Chemistry • Chapter Notes
🌐 vardaanlearning.com📞 9508841336
Module 1: Basic Concepts of Chemistry
Dear Class 12 Student! Welcome to Chemistry! The very first chapter of your Class 12 syllabus is "Solutions", and it is almost entirely based on the concentration terms you are about to revise here (Molarity, Molality, Mole Fraction). Furthermore, your third chapter, "Electrochemistry", requires a flawless understanding of Stoichiometry and Moles. Master this module, and your entry into Class 12 Chemistry will be perfectly smooth!
1. Atomic Mass, Average Atomic Mass, and Molecular Mass
- Atomic Mass: The mass of a single atom. It is expressed in Atomic Mass Units (amu) or unified mass ($u$). By definition, $1 \text{ amu} = \frac{1}{12}$th the mass of one Carbon-12 ($^{12}C$) atom.
- Average Atomic Mass: Elements occur in nature as a mixture of isotopes. The average atomic mass takes into account the percentage abundance of each isotope.
Formula: $\text{Avg. Mass} = \frac{(M_1 \times \%_1) + (M_2 \times \%_2) + \dots}{100}$
- Molecular Mass: The sum of atomic masses of all the elements present in a molecule. (e.g., $H_2O = 2(1) + 16 = 18 \text{ u}$).
2. Mole Concept and Molar Mass
A "Mole" is just a number, exactly like a "dozen" means 12. In chemistry, atoms are so small that we need a massive number to represent a measurable quantity.
Concept
1 Mole ($N_A$) = $6.022 \times 10^{23}$ entities (atoms, molecules, ions, or electrons). This is Avogadro's Number.
Molar Mass ($M$): The mass of one entire mole of a substance in grams. It is numerically equal to its atomic/molecular mass in $u$.
Example: Molecular mass of $H_2O$ is $18 \text{ u}$. Molar mass of $H_2O$ is $18 \text{ g/mol}$.
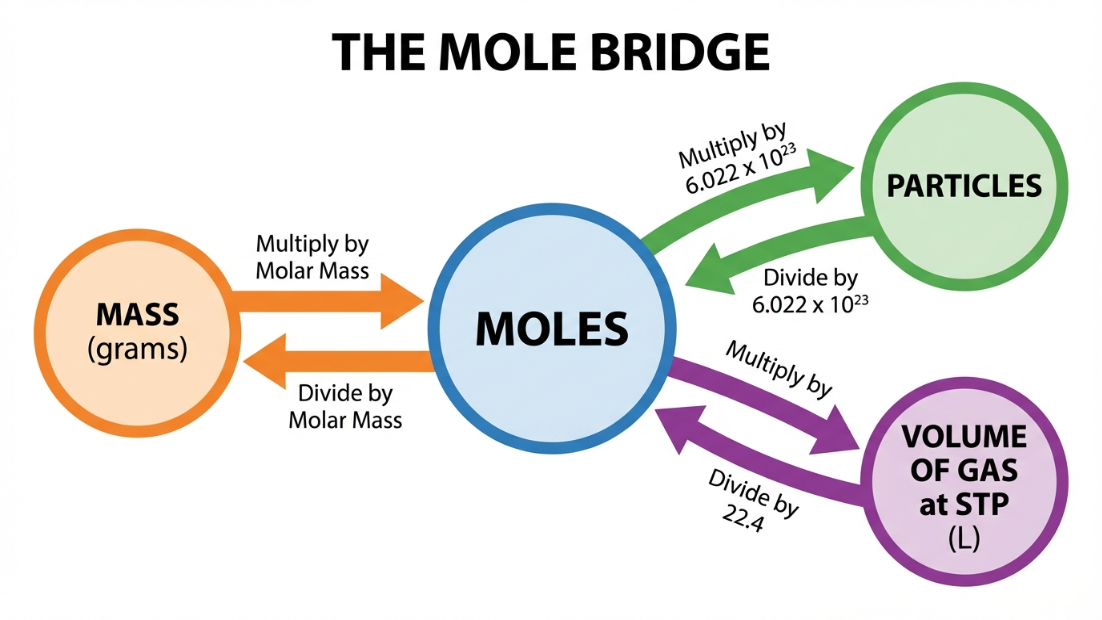
The Golden Formulas
To find the number of moles ($n$):
- From Mass ($w$): $n = \frac{w}{M}$ (where $M$ is Molar Mass)
- From Number of Particles ($N$): $n = \frac{N}{N_A}$
- From Volume of Gas at STP ($V$): $n = \frac{V}{22.4 \text{ Liters}}$
Practice Problem 1
Question: Calculate the number of molecules present in $34.2 \text{ g}$ of cane sugar ($C_{12}H_{22}O_{11}$).
Solution:
1. Find Molar Mass ($M$) of $C_{12}H_{22}O_{11}$:
$M = (12 \times 12) + (22 \times 1) + (11 \times 16) = 144 + 22 + 176 = 342 \text{ g/mol}$.
2. Calculate Moles ($n$):
$n = \frac{w}{M} = \frac{34.2}{342} = \mathbf{0.1 \text{ moles}}$.
3. Calculate Molecules:
$\text{Molecules} = n \times N_A = 0.1 \times 6.022 \times 10^{23} = \mathbf{6.022 \times 10^{22} \text{ molecules}}$.
3. Percentage Composition
This tells us the mass percentage of each element in a given compound.
$$\text{Mass \% of an element} = \frac{\text{Mass of that element in 1 mole of compound}}{\text{Molar mass of compound}} \times 100$$
4. Empirical Formula and Molecular Formula
- Empirical Formula: Represents the simplest whole-number ratio of various atoms present in a compound. (e.g., Benzene's empirical formula is $CH$).
- Molecular Formula: Represents the exact number of different types of atoms present in a molecule. (e.g., Benzene's molecular formula is $C_6H_6$).
Relation: $\text{Molecular Formula} = n \times (\text{Empirical Formula})$, where $n = \frac{\text{Molecular Mass}}{\text{Empirical Formula Mass}}$.
Practice Problem 2
Question: A compound contains $4.07\%$ hydrogen, $24.27\%$ carbon, and $71.65\%$ chlorine. Its molar mass is $98.96 \text{ g}$. What are its empirical and molecular formulas?
Solution:
Step 1: Convert % to grams (assume $100\text{g}$ total): $H = 4.07\text{g}$, $C = 24.27\text{g}$, $Cl = 71.65\text{g}$.
Step 2: Convert grams to moles (divide by atomic mass):
$n_H = \frac{4.07}{1} = 4.07$
$n_C = \frac{24.27}{12} = 2.02$
$n_{Cl} = \frac{71.65}{35.5} = 2.02$
Step 3: Find simplest ratio (divide by smallest value, $2.02$):
$H = \frac{4.07}{2.02} \approx 2$
$C = \frac{2.02}{2.02} = 1$
$Cl = \frac{2.02}{2.02} = 1$
Empirical Formula = $CH_2Cl$
Step 4: Find $n$. Empirical Formula Mass = $12 + 2(1) + 35.5 = 49.5 \text{ g/mol}$.
$n = \frac{98.96}{49.5} \approx 2$.
Molecular Formula = $2 \times (CH_2Cl) = C_2H_4Cl_2$.
5. Stoichiometry and Limiting Reagent
Stoichiometry deals with the calculation of masses (and volumes) of the reactants and products involved in a chemical reaction. A balanced chemical equation is mandatory!
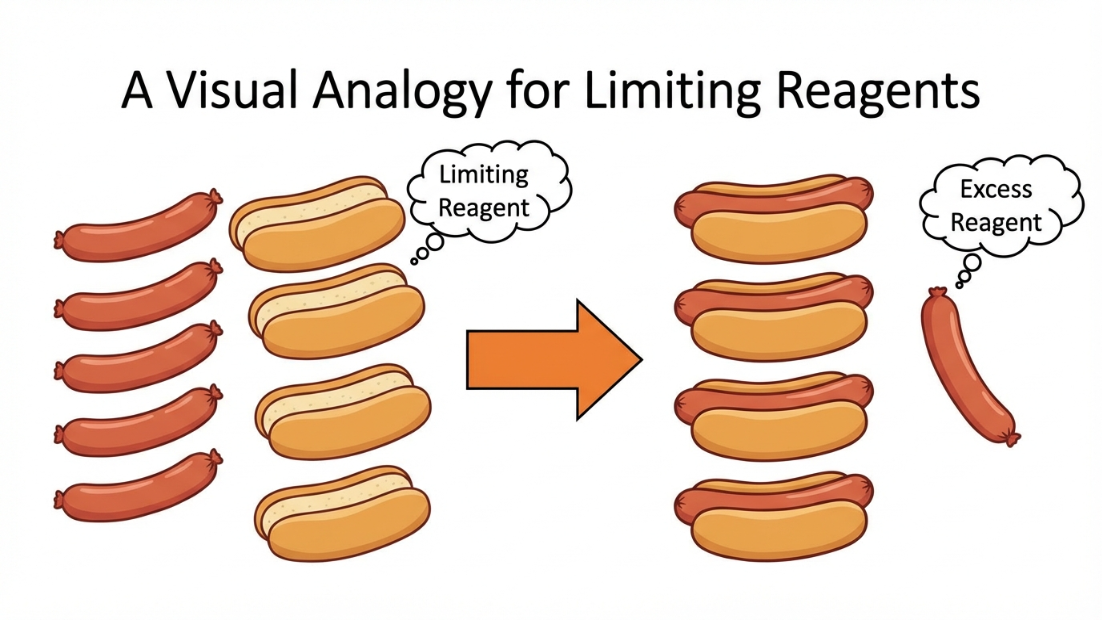
Limiting Reagent: In many reactions, reactants are not present in the exact stoichiometric ratio. The reactant that is completely consumed first limits the amount of product formed. This is the Limiting Reagent (LR).
Trick to find LR
For a balanced reaction: $aA + bB \rightarrow cC$
Calculate the ratio $\frac{\text{Given Moles}}{\text{Stoichiometric Coefficient}}$ for each reactant. (i.e., calculate $\frac{n_A}{a}$ and $\frac{n_B}{b}$).
The reactant with the smallest ratio is the Limiting Reagent! All subsequent product calculations must be based on the LR.
Practice Problem 3
Question: $50.0 \text{ kg}$ of $N_2$ and $10.0 \text{ kg}$ of $H_2$ are mixed to produce $NH_3$. Calculate the mass of $NH_3$ formed. Identify the limiting reagent.
Solution:
1. Balanced Equation: $N_2(g) + 3H_2(g) \rightarrow 2NH_3(g)$
2. Calculate Moles given:
$n_{N_2} = \frac{50,000 \text{ g}}{28 \text{ g/mol}} = 1785.7 \text{ mol}$
$n_{H_2} = \frac{10,000 \text{ g}}{2 \text{ g/mol}} = 5000 \text{ mol}$
3. Find LR (Divide moles by stoichiometric coefficient):
For $N_2$: $\frac{1785.7}{1} = 1785.7$
For $H_2$: $\frac{5000}{3} = 1666.6$
Since $1666.6 < 1785.7$, $H_2$ is the Limiting Reagent.
4. Calculate Product using LR:
From equation, 3 moles of $H_2$ produce 2 moles of $NH_3$.
So, 5000 moles of $H_2$ produce $\frac{2}{3} \times 5000 = 3333.3 \text{ moles of } NH_3$.
5. Convert moles of $NH_3$ to mass:
Mass = $n \times M = 3333.3 \times 17 \text{ g/mol} = 56666 \text{ g} = \mathbf{56.67 \text{ kg}}$.
6. Methods of Expressing Concentration of Solutions
This section is the absolute foundation for Class 12 Chapter 1 (Solutions). Pay very close attention to the formulas and which ones depend on temperature.
A. Mass Percentage (w/w)
$$\text{Mass \% of component A} = \frac{\text{Mass of component A in solution}}{\text{Total mass of the solution}} \times 100$$
B. Volume Percentage (v/v)
$$\text{Volume \% of component A} = \frac{\text{Volume of component A}}{\text{Total volume of solution}} \times 100$$
C. Mole Fraction ($x$ or $\chi$)
The ratio of number of moles of a particular component to the total number of moles of the solution. For a binary solution with components A (solvent) and B (solute):
$$x_A = \frac{n_A}{n_A + n_B} \quad \text{and} \quad x_B = \frac{n_B}{n_A + n_B}$$
Important Property: The sum of mole fractions of all components is always 1 ($x_A + x_B = 1$). Mole fraction is unitless and temperature-independent.
D. Molarity ($M$)
It is defined as the number of moles of solute dissolved in one Litre (or $1 \text{ dm}^3$) of the solution.
Formula
$$\text{Molarity } (M) = \frac{\text{Moles of Solute } (n)}{\text{Volume of Solution in Litres } (V_L)}$$
Or, $M = \frac{w \times 1000}{M_{\text{solute}} \times V_{ml}}$
E. Molality ($m$)
It is defined as the number of moles of solute dissolved in $1 \text{ kg}$ ($1000 \text{ g}$) of the SOLVENT.
Formula
$$\text{Molality } (m) = \frac{\text{Moles of Solute } (n)}{\text{Mass of Solvent in kg } (W_{kg})}$$
Or, $m = \frac{w \times 1000}{M_{\text{solute}} \times W_{\text{solvent in grams}}}$
Crucial Class 12 Board Concept
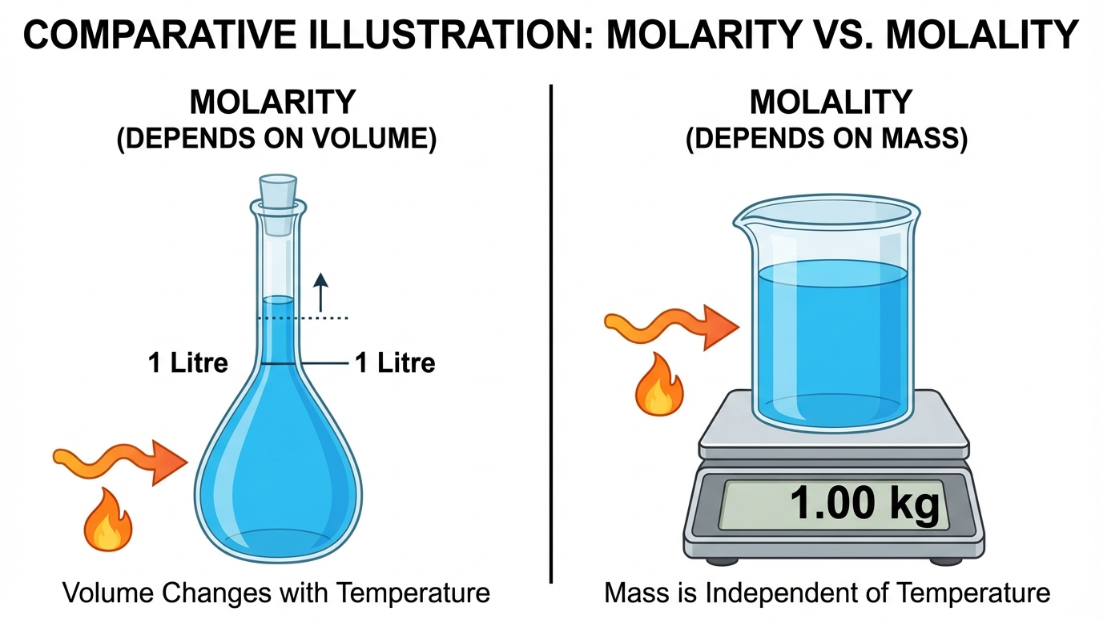
Why is Molality ($m$) preferred over Molarity ($M$) in scientific experiments?
Molarity depends on the Volume of the solution. Since liquids expand or contract with temperature changes, the volume changes, and thus Molarity changes with temperature.
Molality depends on the Mass of the solvent. Mass does not change with temperature. Therefore, Molality is Temperature Independent.
Practice Problem 4
Question: A solution is prepared by dissolving $4 \text{ g}$ of $NaOH$ to give $500 \text{ mL}$ of it. If the density of the solution is $1.02 \text{ g/mL}$, calculate the Molarity and Molality of the solution.
Solution:
Given: Mass of solute ($w$) = $4 \text{ g}$. Molar mass of $NaOH$ ($M$) = $40 \text{ g/mol}$. Volume of sol ($V$) = $500 \text{ mL}$. Density ($d$) = $1.02 \text{ g/mL}$.
1. Molarity ($M$):
Moles of $NaOH = \frac{4}{40} = 0.1 \text{ mol}$.
Volume in L = $0.5 \text{ L}$.
$M = \frac{0.1}{0.5} = \mathbf{0.2 \text{ M}}$ (or $0.2 \text{ mol/L}$).
2. Molality ($m$):
To find molality, we need the mass of the solvent.
Mass of Solution = Volume $\times$ Density = $500 \text{ mL} \times 1.02 \text{ g/mL} = 510 \text{ g}$.
Mass of Solvent = Mass of Solution - Mass of Solute = $510 \text{ g} - 4 \text{ g} = 506 \text{ g} = 0.506 \text{ kg}$.
$m = \frac{\text{Moles of solute}}{\text{Mass of solvent (kg)}} = \frac{0.1}{0.506} \approx \mathbf{0.198 \text{ m}}$ (or $0.198 \text{ mol/kg}$).