Vardaan Learning Institute
Class 11 Chemistry • Chapter Notes
🌐 vardaanlearning.com📞 9508841336
Module 2: Structure of Atom & Periodicity
Dear Class 12 Student! Do not skip this! Chapters like "d- and f-Block Elements" and "Coordination Compounds" in Class 12 completely depend on your ability to write correct electronic configurations (especially for transition metal ions like $Fe^{2+}$ and $Cu^{2+}$) and visualizing the exact shapes of $d$-orbitals. If you don't know the rules for filling electrons and the concept of shielding, advanced theories in Class 12 will make zero sense. Let's lock in these concepts in extreme detail.
1. Quantum Numbers ($n, l, m_l, m_s$)
An electron in an atom is completely designated by a set of four numbers known as Quantum Numbers. Think of them as the precise "postal address" of an electron: State, City, Street, and House Number.
The 4 Numbers
- Principal Quantum Number ($n$): Denotes the main shell. Determines the size and main energy of the orbital. $n = 1, 2, 3, 4\dots$ (K, L, M, N shells).
- Azimuthal / Angular Momentum ($l$): Denotes the subshell. Determines the 3D shape of the orbital and the orbital angular momentum ($L = \sqrt{l(l+1)}\frac{h}{2\pi}$).
For a given $n$, $l$ ranges from $0$ to $(n-1)$.
If $l=0$ (s), $l=1$ (p), $l=2$ (d), $l=3$ (f).
- Magnetic Quantum Number ($m_l$): Denotes the spatial orientation of the orbital in a magnetic field.
For a given $l$, $m_l$ ranges from $-l$ to $+l$ (including zero). Total values = $2l + 1$.
- Spin Quantum Number ($m_s$): Denotes the intrinsic spin of the electron on its own axis. Can only be $+\frac{1}{2}$ (spin up) or $-\frac{1}{2}$ (spin down).
The Concept of Nodes
A node is a region in space around the nucleus where the probability of finding an electron is exactly zero ($|\psi|^2 = 0$). There are two types of nodes:
- Radial (Spherical) Nodes: Spherical surfaces where probability is zero. Formula: $n - l - 1$
- Angular Nodes (Nodal Planes): Flat planes passing through the nucleus where probability is zero. Formula: $l$
- Total Nodes: $(n - l - 1) + l = n - 1$
Practice Problem 1
Question: Calculate the number of radial nodes, angular nodes, and total nodes for a $4d$ orbital. What are the possible values of $m_l$?
Solution:
For a $4d$ orbital, $n = 4$ and $l = 2$ (since it is a 'd' subshell).
1. Radial Nodes: $n - l - 1 = 4 - 2 - 1 = \mathbf{1 \text{ radial node}}$.
2. Angular Nodes: $l = \mathbf{2 \text{ angular nodes}}$ (nodal planes).
3. Total Nodes: $n - 1 = 4 - 1 = \mathbf{3 \text{ total nodes}}$.
4. Possible $m_l$ values: Since $l=2$, $m_l$ can be $\mathbf{-2, -1, 0, +1, +2}$ (representing the 5 distinct d-orbitals).
2. Shapes of s, p, and d Orbitals
An orbital is a 3D boundary surface within which the probability of finding an electron is very high (about 90-95%).
- s-orbital ($l=0$): Spherically symmetrical. Non-directional. For a $1s$ orbital, the electron density is highest at the nucleus. For $2s$, there is one radial node (an empty spherical shell) inside.
- p-orbital ($l=1$): Dumbbell-shaped. Has directional properties along the Cartesian axes. The three p-orbitals are mutually perpendicular ($p_x, p_y, p_z$). The nucleus lies at the nodal plane where electron density is zero.
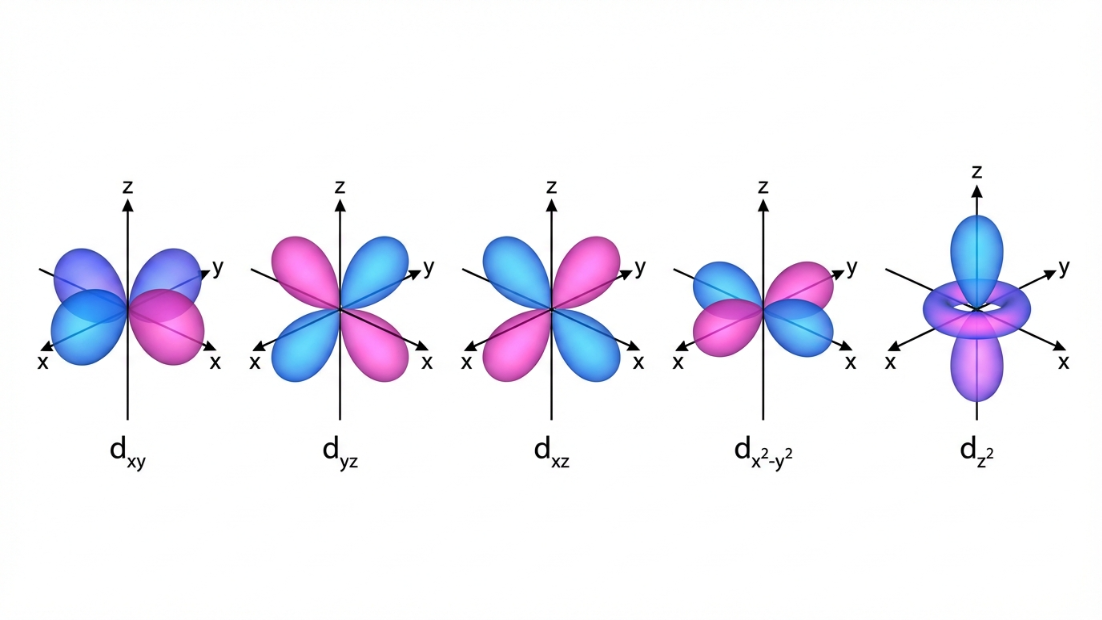
- d-orbital ($l=2$): Double-dumbbell shaped (complex shapes). There are 5 degenerate (equal energy) d-orbitals in an isolated atom:
- $d_{xy}, d_{yz}, d_{zx}$: The four lobes lie between the axes. Nodal planes are the standard axis planes.
- $d_{x^2-y^2}$: The four lobes lie exactly along the x and y axes.
- $d_{z^2}$: A unique shape with two lobes along the z-axis and a high-density electron "doughnut" ring in the xy-plane.
Class 12 Crystal Field Theory Prep
In Coordination Compounds (Class 12), negatively charged ligands approach the central metal ion along the x, y, and z axes. Because the $d_{x^2-y^2}$ and $d_{z^2}$ orbitals point directly at these ligands, their electrons experience massive repulsion, splitting the d-orbitals into two distinct energy levels! Visualizing this geometry is paramount.
3. Rules for Filling Electrons in Orbitals
To determine the multi-electron configuration of an atom, we apply three strict rules:
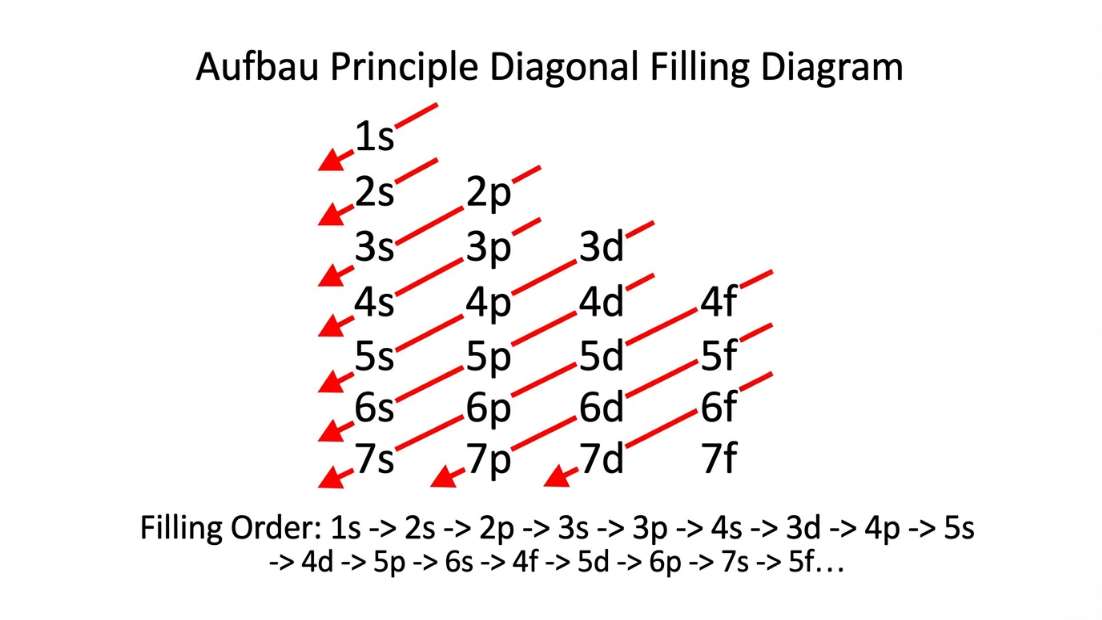
A. Aufbau Principle
"In the ground state of atoms, orbitals are filled in order of their increasing energies."
The $(n+l)$ Rule (Bohr-Bury Rule): The orbital with a lower value of $(n+l)$ has lower energy. If two orbitals happen to have the exact same $(n+l)$ value, the orbital with the lower principal quantum number ($n$) is filled first.
| Orbital |
$n$ |
$l$ |
$(n+l)$ Value |
Filling Order |
| 4s |
4 |
0 |
4 |
Fills First (Lower $n+l$) |
| 3d |
3 |
2 |
5 |
Fills Second |
| 4p |
4 |
1 |
5 |
Fills Third (Same $n+l$, but higher $n$) |
B. Pauli Exclusion Principle
"No two electrons in an atom can have the same set of four quantum numbers."
Consequence: An orbital can hold a maximum of two electrons, and they must have opposite spins ($\uparrow \downarrow$). If they had the same spin, all four quantum numbers would be identical, which nature forbids.
C. Hund's Rule of Maximum Multiplicity
"Electron pairing in degenerate orbitals (orbitals of the same subshell, like $p_x, p_y, p_z$) will not take place until each orbital of that subshell is singly occupied with parallel spins."
Electrons repel each other. By occupying different orbitals first with parallel spins, they minimize repulsion and maximize "Exchange Energy," leading to a more stable atom.
4. Electronic Configuration (1-30 & The Great Exceptions)
Following the Aufbau order ($1s < 2s < 2p < 3s < 3p < 4s < 3d$), let's look at standard configurations and the extremely important exceptions.
Standard Examples
- Carbon ($Z=6$): $1s^2 2s^2 2p^2$
- Chlorine ($Z=17$): $1s^2 2s^2 2p^6 3s^2 3p^5$ or $[Ne] 3s^2 3p^5$
- Iron ($Z=26$): $[Ar] 4s^2 3d^6$
Crucial Exceptions (Class 12 Boards Favorite)
Chromium (Cr, $Z=24$):
Expected: $[Ar] 4s^2 3d^4$ $\rightarrow$ Actual: $[Ar] 4s^1 3d^5$
Copper (Cu, $Z=29$):
Expected: $[Ar] 4s^2 3d^9$ $\rightarrow$ Actual: $[Ar] 4s^1 3d^{10}$
Why does an electron jump from 4s to 3d? Two primary reasons:
1. Symmetrical Distribution: Half-filled ($d^5$) and fully-filled ($d^{10}$) subshells are perfectly symmetrical, leading to less electron shielding and greater nuclear attraction.
2. Exchange Energy: Electrons with parallel spins in degenerate orbitals can exchange positions. The more parallel spins, the more possible exchanges. Each exchange releases energy (Exchange Energy). A $d^5$ configuration has 10 possible exchanges, making it highly thermodynamically stable.
Writing Configurations for Transition Metal Ions (The d-Block Trap)
When transition metals (d-block) lose electrons to form positive ions, electrons are ALWAYS removed from the outermost principal shell ($ns$) before the inner $(n-1)d$ shell. Even though $4s$ filled before $3d$, once the $3d$ starts filling, the $4s$ electrons are physically further away from the nucleus!
Practice Problem 2
Question: Write the electronic configuration for $Fe$, $Fe^{2+}$, and $Fe^{3+}$. ($Z = 26$)
Solution:
1. Neutral Iron ($Fe$):
Filling order: $[Ar] 4s^2 3d^6$.
2. Iron(II) Ion ($Fe^{2+}$):
We must remove 2 electrons. They leave the outermost shell ($4s$) first!
Configuration: $\mathbf{[Ar] 3d^6}$ (or $[Ar] 4s^0 3d^6$).
3. Iron(III) Ion ($Fe^{3+}$):
Remove 1 more electron. Now that $4s$ is empty, we take from the $3d$.
Configuration: $\mathbf{[Ar] 3d^5}$.
(Notice that $Fe^{3+}$ is much more stable than $Fe^{2+}$ because it achieves the highly coveted half-filled $d^5$ configuration!)
5. Periodic Trends in Properties
Understanding these trends—and specifically the concept of shielding—is non-negotiable for mastering the p-block, d-block, and f-block chapters in Class 12.
Concept: Effective Nuclear Charge ($Z_{eff}$) and Shielding Effect
In a multi-electron atom, the outer electrons do not feel the full positive charge of the nucleus ($Z$). The inner core electrons "shield" or "screen" the outer electrons from the nucleus. The actual net positive pull felt by an outer electron is the Effective Nuclear Charge ($Z_{eff}$).
$Z_{eff} = Z - \sigma$ (where $\sigma$ is the shielding constant).
Crucial Fact: The shielding ability of subshells drops drastically in the order: $s > p > d > f$. Because d and f orbitals are so diffuse (spread out), they are terrible at shielding. This poor shielding by d/f electrons causes the famous "Lanthanoid Contraction" in Class 12.
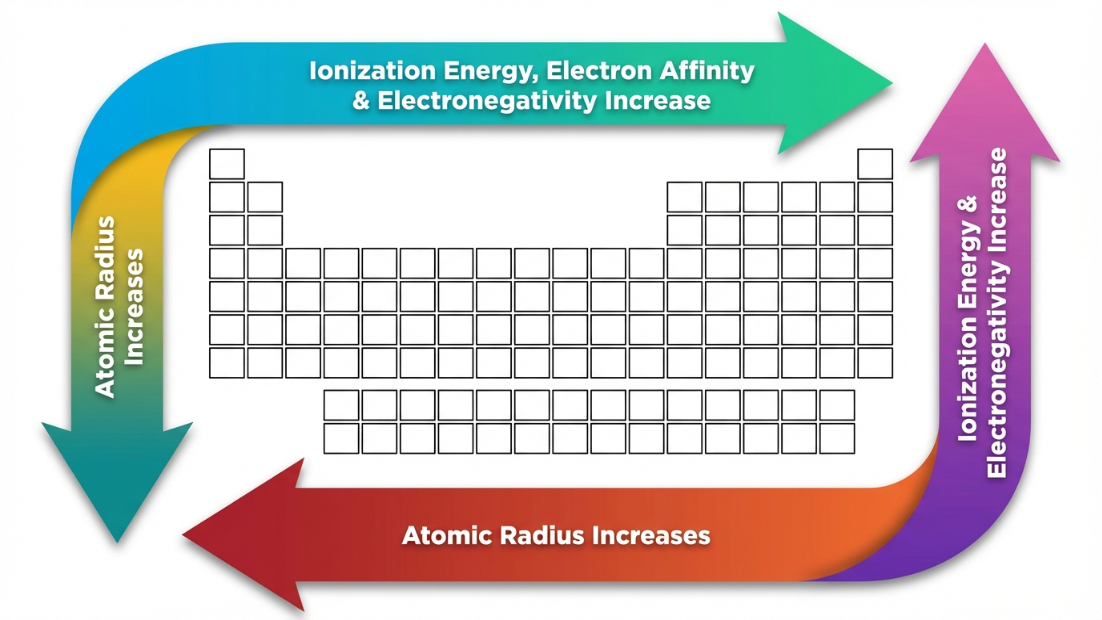
A. Atomic and Ionic Radii
- Across a Period (Left to Right): Decreases. Electrons are added to the same shell, but protons are added to the nucleus. $Z_{eff}$ increases, pulling the electron cloud tighter.
- Down a Group (Top to Bottom): Increases. Entirely new principal shells are added, increasing the distance from the nucleus. The shielding effect dominates over the increased nuclear charge.
- Ions: A Cation ($Na^+$) is always much smaller than its parent atom (loss of entire outer shell, higher $Z_{eff}$ per remaining electron). An Anion ($Cl^-$) is always larger than its parent atom (added electron increases electron-electron repulsion, causing the cloud to expand).
B. Ionization Enthalpy ($\Delta_i H$)
The minimum energy required to remove the most loosely bound electron from an isolated gaseous atom in its ground state. $M(g) \rightarrow M^+(g) + e^-$
- Trend: Generally increases across a period (higher $Z_{eff}$, harder to remove) and decreases down a group (larger radius, easier to remove).
- Important Exceptions:
- Nitrogen ($1s^2 2s^2 2p^3$) > Oxygen ($1s^2 2s^2 2p^4$): N has a perfectly half-filled $2p^3$ configuration, making it exceptionally stable and harder to ionize than O.
- Beryllium ($1s^2 2s^2$) > Boron ($1s^2 2s^2 2p^1$): The $2s$ electron in Be is more penetrating (closer to nucleus) and fully filled compared to the lone $2p$ electron in B.
- Successive Ionization Enthalpies: The second IE is always higher than the first ($IE_2 > IE_1$). A massive "jump" in successive IE values indicates that a stable noble gas core has been broken into. (e.g., Sodium has a low $IE_1$, but an astronomically high $IE_2$).
C. Electron Gain Enthalpy ($\Delta_{eg} H$)
The enthalpy change when an electron is added to an isolated gaseous atom. Can be exothermic (negative) or endothermic (positive).
- Trend: Becomes more negative (more energy released) as we move left to right. Halogens (Group 17) are one electron short of a noble gas configuration and have the most negative values.
- Noble Gases: Have large positive electron gain enthalpies because their shells are full. Adding an electron forces it into a higher, unstable principal shell, requiring energy input.
- The Big Exception: Chlorine (Cl) has a MORE negative electron gain enthalpy than Fluorine (F).
Reason: Fluorine is extremely small. The added electron suffers severe inter-electronic repulsion in the highly compact $2p$ subshell. Chlorine's larger $3p$ subshell accommodates the new electron much more comfortably.
D. Electronegativity
A qualitative measure of the ability of an atom within a chemical bond to attract the shared pair of electrons towards itself. Unlike EGE (which is for isolated atoms), EN applies to bonded atoms.
- Trend: Increases across a period, decreases down a group.
- Fact: Fluorine (F) is the most electronegative element in the periodic table (value = 4.0 on Pauling scale), followed by Oxygen (3.5) and Nitrogen (3.0). Understanding this dictates all of Organic Chemistry dipole moments in Class 12!
Practice Problem 3
Question: Arrange the following in decreasing order of ionic radii: $N^{3-}, O^{2-}, F^-, Na^+, Mg^{2+}, Al^{3+}$.
Solution:
First, observe the number of electrons in each ion:
$N^{3-}$ (7+3=10e), $O^{2-}$ (8+2=10e), $F^-$ (9+1=10e), $Na^+$ (11-1=10e), $Mg^{2+}$ (12-2=10e), $Al^{3+}$ (13-3=10e).
These are Isoelectronic species (they all have exactly 10 electrons).
For isoelectronic species, the ionic radius depends entirely on the nuclear charge (number of protons, $Z$).
Protons: N(7), O(8), F(9), Na(11), Mg(12), Al(13).
The higher the nuclear charge (more protons), the stronger the inward pull on the 10 electrons, and the smaller the radius.
Decreasing order of size (least protons to most protons):
$\mathbf{N^{3-} > O^{2-} > F^- > Na^+ > Mg^{2+} > Al^{3+}}$
Practice Problem 4
Question: The successive ionization enthalpies of an element 'X' are $738$, $1450$, $7730$, and $10500 \text{ kJ/mol}$. To which group of the periodic table does element 'X' belong?
Solution:
Look for the massive "jump" in energy values.
$IE_1 = 738$
$IE_2 = 1450$ (About double, normal increase)
$IE_3 = 7730$ (Massive 5x jump!)
$IE_4 = 10500$
The enormous jump between $IE_2$ and $IE_3$ means that the third electron is being removed from a highly stable, completely filled noble gas core. This implies the atom only had 2 loosely bound valence electrons. Therefore, element 'X' belongs to Group 2 (Alkaline Earth Metals). (Specifically, these values match Magnesium).