Vardaan Learning Institute
Class 11 Chemistry • Chapter Notes
🌐 vardaanlearning.com📞 9508841336
Module 3: Chemical Bonding and Molecular Structure
Dear Class 12 Student! This module is the absolute backbone of Organic Chemistry. If you cannot identify the hybridization ($sp, sp^2, sp^3$) of a carbon atom, you will not understand organic reaction mechanisms, electrophilic attacks, or resonance. Furthermore, concepts like Hydrogen Bonding dictate boiling point trends and solubility in almost every p-block and organic chapter. Master these foundational tools now!
1. Ionic and Covalent Bonding
Atoms combine to achieve a stable octet (8 electrons in their valence shell), similar to noble gases.
- Ionic (Electrovalent) Bond: Formed by the complete transfer of one or more electrons from an electropositive atom (metal) to an electronegative atom (non-metal). Example: $NaCl$. They form crystal lattices, not discrete molecules.
- Covalent Bond: Formed by the mutual sharing of electrons between two atoms (usually non-metals). Example: $H_2O, CH_4$.
2. Lewis Dot Structures and Formal Charge
Lewis structures show the valence electrons as dots around the atomic symbol. Sometimes, the total charge of a polyatomic ion is distributed unevenly among its atoms. We calculate the Formal Charge (FC) to find the most stable structure (the one with FCs closest to zero).
Formula
Formal Charge ($FC$):
$$FC = V - N - \frac{B}{2}$$
Where:
- $V$ = Total number of valence electrons in the free atom.
- $N$ = Total number of non-bonding (lone pair) electrons.
- $B$ = Total number of bonding (shared) electrons.
Practice Problem 1
Question: Calculate the formal charge on each oxygen atom in the Ozone molecule ($O_3$).
Solution:
Draw the Lewis structure of Ozone: $\ddot{O}=\ddot{O}^+-\ddot{\ddot{O}}^-$
Let's number them: $O_1$ (double bonded), $O_2$ (central), $O_3$ (single bonded). All oxygen atoms have $V = 6$.
For $O_1$: $N = 4$ (two lone pairs), $B = 4$ (double bond).
$FC = 6 - 4 - \frac{4}{2} = 6 - 4 - 2 = \mathbf{0}$
For $O_2$ (central): $N = 2$ (one lone pair), $B = 6$ (one double, one single bond).
$FC = 6 - 2 - \frac{6}{2} = 6 - 2 - 3 = \mathbf{+1}$
For $O_3$: $N = 6$ (three lone pairs), $B = 2$ (single bond).
$FC = 6 - 6 - \frac{2}{2} = 6 - 6 - 1 = \mathbf{-1}$
Check: Sum of FCs = $0 + 1 - 1 = 0$, which matches the neutral charge of the $O_3$ molecule.
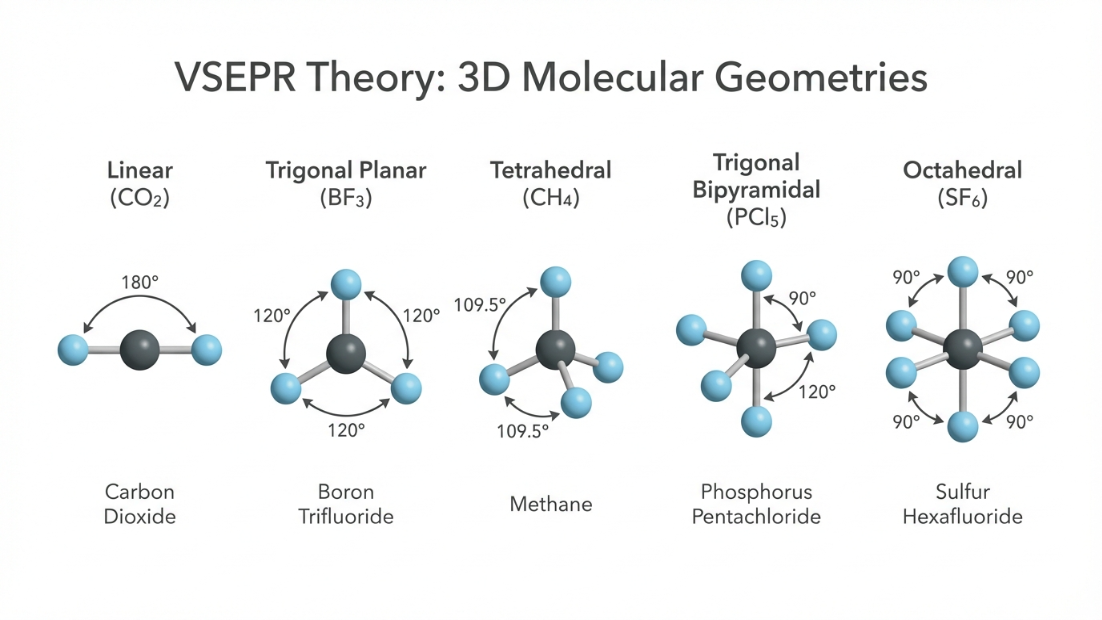
3. Valence Shell Electron Pair Repulsion (VSEPR) Theory
Lewis structures do not tell us the 3D shape of a molecule. VSEPR theory states that electron pairs around a central atom will arrange themselves to minimize repulsion and maximize distance between them.
VSEPR Rules
The repulsive interaction of electron pairs decreases in the order:
Lone Pair - Lone Pair (lp-lp) > Lone Pair - Bond Pair (lp-bp) > Bond Pair - Bond Pair (bp-bp)
Class 12 Trap: A lone pair is "fatter" and closer to the central atom, taking up more space and pushing bond pairs closer together, which reduces standard bond angles (e.g., $109.5^\circ$ in $CH_4$ drops to $104.5^\circ$ in $H_2O$ due to two lone pairs).
| Total Electron Pairs |
Geometry |
Bond Angle |
Example |
| 2 |
Linear |
$180^\circ$ |
$BeCl_2$, $CO_2$ |
| 3 |
Trigonal Planar |
$120^\circ$ |
$BF_3$ |
| 4 |
Tetrahedral |
$109.5^\circ$ |
$CH_4$ |
| 5 |
Trigonal Bipyramidal |
$90^\circ$, $120^\circ$ |
$PCl_5$ |
| 6 |
Octahedral |
$90^\circ$ |
$SF_6$ |
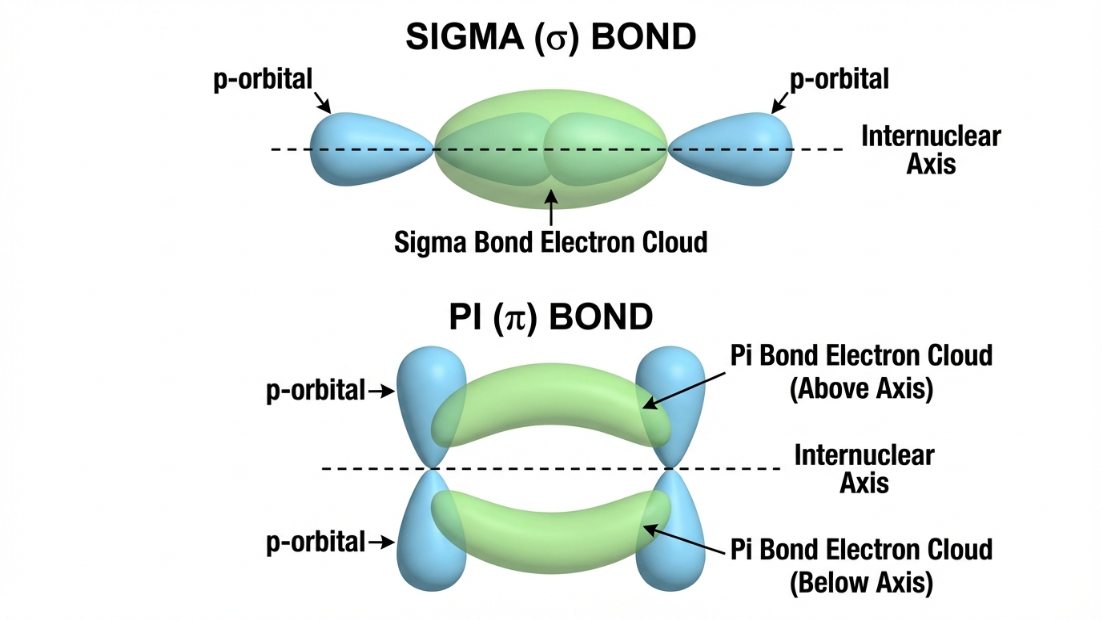
4. Valence Bond Theory (VBT): Sigma ($\sigma$) and Pi ($\pi$) Bonds
VBT states that covalent bonds are formed by the overlapping of half-filled atomic orbitals.
- Sigma ($\sigma$) Bond: Formed by head-on (axial) overlap of orbitals (s-s, s-p, or p-p). The electron density is concentrated directly between the two nuclei. It is a strong bond, and allows free rotation around the bond axis.
- Pi ($\pi$) Bond: Formed by sideways (lateral) overlap of parallel p-orbitals. The electron density is above and below the internuclear axis. It is a weaker bond, and restricts rotation.
Organic Chemistry Alert
In organic chemistry, a single bond is always a $\sigma$ bond. A double bond consists of $1\sigma + 1\pi$. A triple bond consists of $1\sigma + 2\pi$.
Because $\pi$ bonds are weaker and their electron clouds are exposed above and below the plane, they are the primary site of attack for Electrophiles (electron-loving species) in alkenes and alkynes!
5. Concept of Hybridization ($sp, sp^2, sp^3...$)
When atoms form bonds, pure atomic orbitals (like 2s and 2p in Carbon) often don't have the right geometry or energy to form symmetrical molecules. Hybridization is the intermixing of slightly different atomic orbitals to produce an equal number of identical, degenerate "hybrid" orbitals.
The Hybridization Cheat Code
To find the hybridization of any central atom, calculate the
Steric Number (SN):
$$SN = (\text{Number of } \sigma \text{ bonds attached}) + (\text{Number of Lone Pairs})$$
- If $SN = 2 \implies \mathbf{sp}$ hybridization (Linear geometry, $180^\circ$)
- If $SN = 3 \implies \mathbf{sp^2}$ hybridization (Trigonal planar geometry, $120^\circ$)
- If $SN = 4 \implies \mathbf{sp^3}$ hybridization (Tetrahedral geometry, $109.5^\circ$)
- If $SN = 5 \implies \mathbf{sp^3d}$ hybridization (Trigonal bipyramidal)
- If $SN = 6 \implies \mathbf{sp^3d^2}$ hybridization (Octahedral)
Note: $\pi$ bonds are NEVER included in hybrid orbitals; they are formed by unhybridized p-orbitals!
Deep Dive: Carbon Hybridization in Organic Chem
- Alkanes (e.g., Methane $CH_4$): Carbon forms 4 $\sigma$ bonds. $SN = 4$. Hybridization is $\mathbf{sp^3}$. Shape is Tetrahedral.
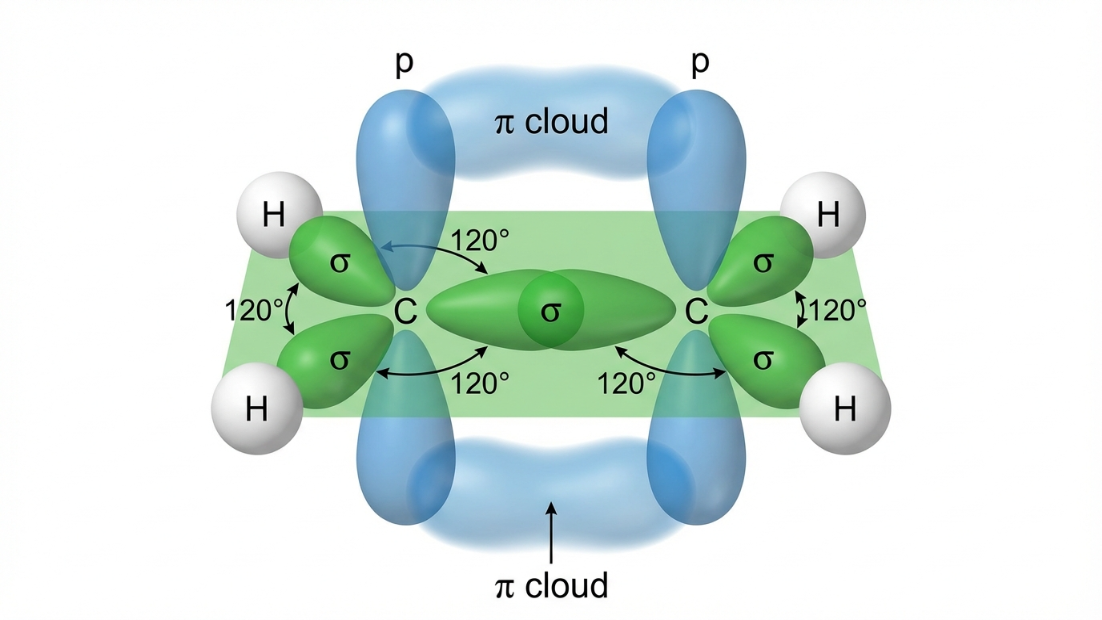
- Alkenes (e.g., Ethene $C_2H_4$): Each Carbon forms 3 $\sigma$ bonds and 1 $\pi$ bond. $SN = 3$. Hybridization is $\mathbf{sp^2}$. Shape is Trigonal Planar. The one unhybridized p-orbital forms the $\pi$ bond.
- Alkynes (e.g., Ethyne $C_2H_2$): Each Carbon forms 2 $\sigma$ bonds and 2 $\pi$ bonds. $SN = 2$. Hybridization is $\mathbf{sp}$. Shape is Linear. The two unhybridized p-orbitals form the two $\pi$ bonds.
Practice Problem 2
Question: Determine the hybridization and shape of the $NH_3$ (Ammonia) molecule.
Solution:
1. Central atom is Nitrogen (N). It has 5 valence electrons.
2. It forms 3 $\sigma$ bonds with 3 Hydrogen atoms (using 3 electrons).
3. This leaves 2 electrons, which form 1 Lone Pair.
4. Steric Number ($SN$) = $\sigma \text{ bonds} + \text{Lone Pairs} = 3 + 1 = 4$.
5. Since $SN = 4$, the hybridization is $\mathbf{sp^3}$.
6. Shape: The base electron geometry is tetrahedral. However, because one position is occupied by an invisible lone pair, the molecular shape is Trigonal Pyramidal with a bond angle of $\approx 107^\circ$.
6. Dipole Moment and Polarity of Molecules
In covalent bonds between different atoms, the more electronegative atom pulls the shared electrons closer, creating partial charges ($\delta+$ and $\delta-$). This creates a bond dipole.
Dipole Moment ($\vec{\mu}$): A vector quantity measuring the polarity of a molecule. It points from the positive center to the negative center.
$$\mu = Q \times r$$
(where $Q$ is magnitude of charge, $r$ is distance of separation). Unit is Debye ($D$).
Crucial Rule: A molecule can have polar bonds but still be a non-polar molecule ($\mu = 0$) if it is perfectly symmetrical, because the individual bond dipole vectors cancel each other out (e.g., $CO_2$, $CCl_4$, $BF_3$).
Practice Problem 3
Question: Why is the dipole moment of $NH_3$ ($1.47 \text{ D}$) much higher than that of $NF_3$ ($0.23 \text{ D}$), even though F is more electronegative than H?
Solution:
Both molecules have a pyramidal shape with one lone pair on Nitrogen. Dipole moment is a vector sum.
In $NH_3$: Nitrogen is more electronegative than Hydrogen. The three N-H bond dipoles point towards Nitrogen. The orbital dipole of the lone pair also points upwards (away from the nucleus). All vectors point in the same general direction, adding up to a large net dipole.
In $NF_3$: Fluorine is more electronegative than Nitrogen. The three N-F bond dipoles point downwards, away from Nitrogen. The orbital dipole of the lone pair points upwards. These vectors oppose each other, partially canceling out, resulting in a very low net dipole moment.
7. Hydrogen Bonding
Hydrogen bonding is a special, unusually strong type of dipole-dipole attraction. It occurs ONLY when Hydrogen is covalently bonded to a highly electronegative, small atom—specifically Fluorine (F), Oxygen (O), or Nitrogen (N).
Because F, O, and N pull the electron cloud so strongly, the tiny Hydrogen nucleus is left highly exposed (a strong $\delta+$ charge), which powerfully attracts the lone pairs of adjacent molecules.
- Intermolecular H-bonding: Between different molecules (e.g., between two $H_2O$ molecules, or between $HF$ molecules). This heavily increases boiling points.
- Intramolecular H-bonding: Occurs within the same molecule (e.g., o-nitrophenol). This forms a ring structure and typically decreases boiling points compared to isomers with intermolecular bonding.
Class 12 Organic Chemistry Application
Why do Alcohols ($R-OH$) have much higher boiling points than Haloalkanes ($R-X$) or Ethers ($R-O-R$) of comparable mass?
Answer: Alcohols form extensive intermolecular Hydrogen bonds due to the -OH group, requiring massive amounts of thermal energy to break the molecules apart. Ethers and Haloalkanes cannot form H-bonds with themselves. This concept will be tested in every organic chapter!