Vardaan Learning Institute
Class 11 Chemistry • Chapter Notes
🌐 vardaanlearning.com📞 9508841336
Module 4: Redox Reactions
Dear Class 12 Student! Chapter 3 of your Class 12 syllabus is Electrochemistry. Every single battery, fuel cell, and electrolysis reaction you will study operates on the principles of Redox. Furthermore, identifying disproportionation reactions is a guaranteed 3-mark question in p-block chemistry. If you cannot calculate structural oxidation states or use the Nernst Equation basics, you will struggle. Let's build an unbreakable foundation!
1. Concept of Oxidation and Reduction
Originally, oxidation meant the addition of oxygen, and reduction meant the removal of oxygen. However, the modern, universally applicable definition is based entirely on electron transfer.
Concept: OIL RIG
- Oxidation Is Loss of electrons. (Oxidation Number increases/becomes more positive)
- Reduction Is Gain of electrons. (Oxidation Number decreases/becomes more negative)
Alternative Mnemonic: LEO (Lose Electrons Oxidation) says GER (Gain Electrons Reduction).
A Redox (Reduction-Oxidation) reaction is one where both oxidation and reduction occur simultaneously. One substance gives electrons; another must take them.
2. Rules for Calculating Oxidation Number (O.N.)
The Oxidation Number (or Oxidation State) is a theoretical charge assigned to an atom in a molecule, assuming all bonds are completely ionic (the more electronegative atom takes both electrons in a bond).
The Golden Rules
- Free State: The O.N. of an element in its free/uncombined state is exactly ZERO. (e.g., $Na$, $H_2$, $O_2$, $P_4$, $S_8$ all have O.N. = $0$).
- Monatomic Ions: The O.N. equals the charge on the ion. (e.g., $Na^+$ is $+1$, $S^{2-}$ is $-2$).
- Oxygen: Almost always $-2$.
Exceptions: Peroxides like $H_2O_2$ (O.N. = $-1$), Superoxides like $KO_2$ (O.N. = $-1/2$), and with Fluorine like $OF_2$ (O.N. = $+2$).
- Hydrogen: Almost always $+1$ when bonded to non-metals.
Exception: When bonded to active metals (hydrides like $NaH$, $LiAlH_4$), it is $-1$.
- Fluorine: ALWAYS $-1$ in all its compounds (it is the most electronegative element, no exceptions).
- Alkali & Alkaline Earth Metals: Group 1 metals are always $+1$. Group 2 metals are always $+2$.
- Sum Rule: The algebraic sum of all O.N.s in a neutral molecule is ZERO. In a polyatomic ion, the sum equals the net charge of the ion.
The Paradox of Fractional Oxidation States & Structural Exceptions
Sometimes, using the basic rules yields a fraction (e.g., in $Fe_3O_4$, $3x - 8 = 0 \implies x = +8/3$). Electrons cannot be shared in fractions! A fractional oxidation state means you are calculating the average oxidation state of two different atoms of the same element in a mixed compound or a specific structure.
Class 12 Traps: Structural O.N.
1. $Fe_3O_4$ (Magnetite): It is actually a mixture of $FeO$ ($Fe$ is $+2$) and $Fe_2O_3$ ($Fe$ is $+3$). The average is $+8/3$.
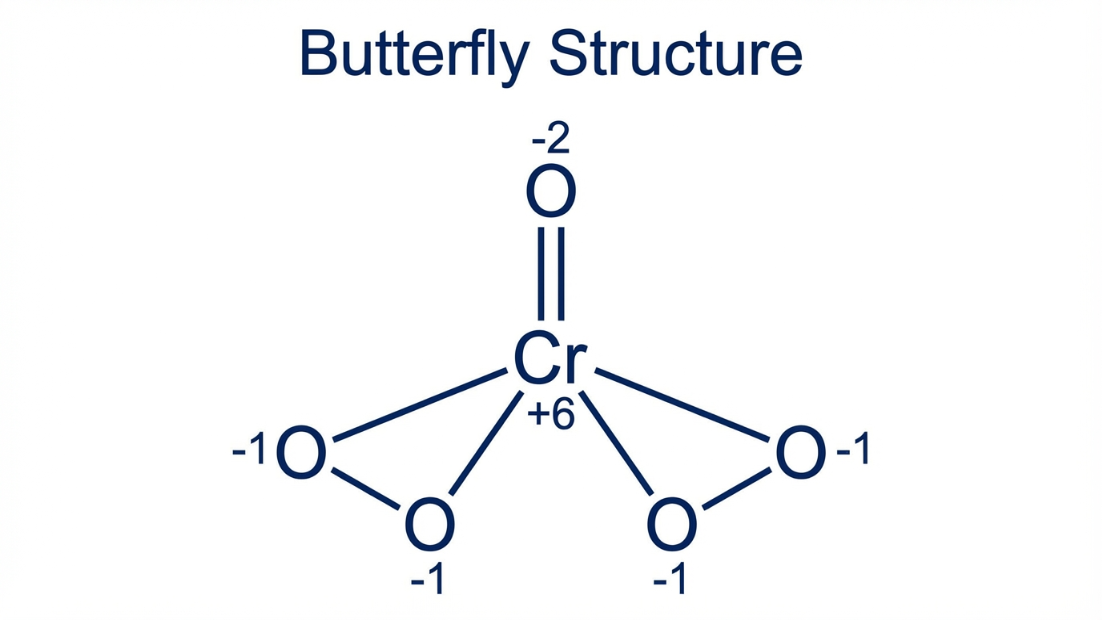
2. $CrO_5$ (Chromium pentoxide): If you use math: $x + 5(-2) = 0 \implies x = +10$. WRONG! Chromium only has 6 valence electrons; it cannot lose 10.
The Reality: $CrO_5$ has a "Butterfly Structure" containing two peroxide linkages (-O-O-). Four oxygens are peroxides (O.N. = -1) and one is a normal double-bonded oxygen (O.N. = -2).
Correct Math: $x + 4(-1) + 1(-2) = 0 \implies \mathbf{x = +6}$.
3. Caro's Acid ($H_2SO_5$): Math gives S = +8 (Wrong, S max is +6). Structurally, it contains one peroxide bond. Correct S O.N. = +6.
3. Types of Redox Reactions
Understanding these patterns helps predict products in Inorganic Chemistry.
- Combination: $A + B \rightarrow C$ (e.g., $C(0) + O_2(0) \rightarrow C(+4)O_2(-2)$)
- Decomposition: $C \rightarrow A + B$ (e.g., $2KClO_3 \rightarrow 2KCl + 3O_2$)
- Displacement: $X + YZ \rightarrow XZ + Y$ (e.g., $Zn + CuSO_4 \rightarrow ZnSO_4 + Cu$)
- Disproportionation: The most important type! A reaction where the same element is simultaneously oxidized and reduced. The element must exist in at least three different oxidation states for this to happen.
Practice Problem 1
Question: Prove that the decomposition of Hydrogen Peroxide ($H_2O_2$) is a disproportionation reaction.
Solution:
Write the reaction: $2H_2O_2(aq) \rightarrow 2H_2O(l) + O_2(g)$
Assign Oxidation Numbers to the Oxygen atoms:
In $H_2O_2$ (a peroxide), O is $-1$.
In $H_2O$ (normal oxide), O is $-2$ (Reduction: $-1 \rightarrow -2$).
In $O_2$ (free state), O is $0$ (Oxidation: $-1 \rightarrow 0$).
Since Oxygen in the -1 state is simultaneously undergoing oxidation and reduction, this is a classic disproportionation reaction.
4. Identifying Oxidizing and Reducing Agents
This terminology often confuses students because it feels backwards. Pay close attention!
- Oxidizing Agent (Oxidant): The substance that oxidizes another substance. To do this, it must steal electrons from the other substance. Therefore, the oxidizing agent itself undergoes reduction (gains electrons, O.N. decreases). Examples: $KMnO_4, K_2Cr_2O_7, HNO_3, O_3$.
- Reducing Agent (Reductant): The substance that reduces another substance. To do this, it must give its electrons to the other substance. Therefore, the reducing agent itself undergoes oxidation (loses electrons, O.N. increases). Examples: $LiAlH_4, Na, H_2S, Zn$.
5. Balancing Redox Reactions
Redox reactions cannot be balanced merely by counting atoms; the total number of electrons lost must perfectly equal the total number of electrons gained. There are two main methods.
Method A: The Oxidation Number Method
- Assign O.N. to all elements and identify those undergoing oxidation and reduction.
- Calculate the increase and decrease in O.N. per atom, then multiply by the number of atoms involved.
- Equalize the total increase and total decrease in O.N. by multiplying formulas by suitable integers.
- Balance all other atoms except O and H.
- Balance O by adding $H_2O$, and H by adding $H^+$ (acidic) or $OH^-$ (basic).
Method B: The Half-Reaction (Ion-Electron) Method
This is the most reliable method for complex ionic equations. Let's walk through an advanced example in a Basic Medium.
Practice Problem 2
Question: Balance the following equation in a
basic medium:
$MnO_4^- + I^- \rightarrow MnO_2 + I_2$
Solution:
Step 1: Split into half-reactions
Oxidation: $I^- \rightarrow I_2$
Reduction: $MnO_4^- \rightarrow MnO_2$
Step 2 to 5: Balance each half independently (Treat as Acidic first)
Oxidation Half:
Balance atoms: $2I^- \rightarrow I_2$
Balance charge: Add 2 $e^-$ to RHS.
$2I^- \rightarrow I_2 + 2e^-$ --- (Eq 1)
Reduction Half:
Balance Mn: Already balanced.
Balance O: Add 2 $H_2O$ to RHS. $\implies MnO_4^- \rightarrow MnO_2 + 2H_2O$
Balance H: Add 4 $H^+$ to LHS. $\implies 4H^+ + MnO_4^- \rightarrow MnO_2 + 2H_2O$
Balance Charge: LHS net $= +3$. RHS net $= 0$. Add 3 $e^-$ to LHS.
$3e^- + 4H^+ + MnO_4^- \rightarrow MnO_2 + 2H_2O$ --- (Eq 2)
Step 6: Equalize Electrons & Add
Multiply Eq 1 by 3, and Eq 2 by 2.
$6I^- \rightarrow 3I_2 + 6e^-$
$6e^- + 8H^+ + 2MnO_4^- \rightarrow 2MnO_2 + 4H_2O$
Add them: $6I^- + 8H^+ + 2MnO_4^- \rightarrow 3I_2 + 2MnO_2 + 4H_2O$
Step 7: Convert to Basic Medium (The Crucial Step!)
Since it's a basic medium, $H^+$ cannot exist freely. Add $8OH^-$ to BOTH sides to neutralize the $8H^+$.
$6I^- + (8H^+ + 8OH^-) + 2MnO_4^- \rightarrow 3I_2 + 2MnO_2 + 4H_2O + 8OH^-$
The $(8H^+ + 8OH^-)$ becomes $8H_2O$.
$6I^- + 8H_2O + 2MnO_4^- \rightarrow 3I_2 + 2MnO_2 + 4H_2O + 8OH^-$
Step 8: Simplify
Cancel 4 $H_2O$ from both sides.
Final Answer: $\mathbf{6I^- + 4H_2O + 2MnO_4^- \rightarrow 3I_2 + 2MnO_2 + 8OH^-}$
6. Concept of Standard Electrode Potential (Class 12 Bridge)
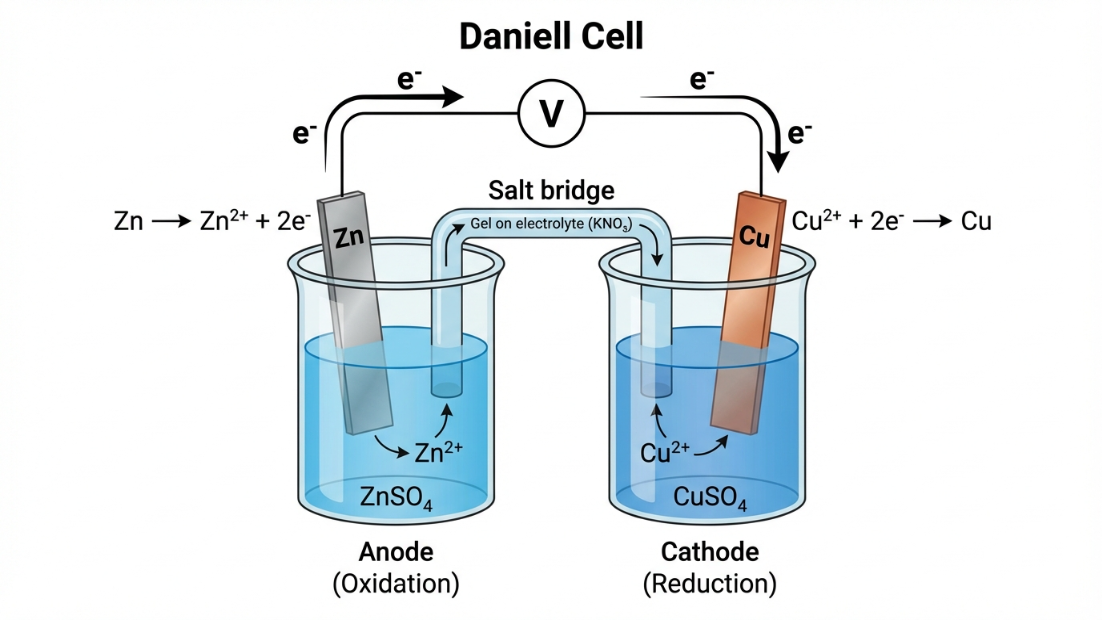
Why does Zinc displace Copper from $CuSO_4$ solution, but Copper cannot displace Zinc from $ZnSO_4$? The answer lies in their inherent "desire" to gain or lose electrons, measured as Electrode Potential.
- When a metal is dipped in a solution of its own ions (e.g., $Zn$ in $Zn^{2+}$), a potential difference develops at the interface.
- By convention, IUPAC uses the Standard Reduction Potential ($E^\circ_{red}$). This measures the tendency of a species to gain electrons and get reduced under standard conditions (Concentration = $1 \text{ M}$, Temperature = $298 \text{ K}$, Pressure = $1 \text{ atm}$).
The Electrochemical Series Rule
The Electrochemical Series (ECS) arranges elements in order of increasing Standard Reduction Potential ($E^\circ$).
The more positive the $E^\circ$, the stronger the species' desire to be reduced.
A species with a higher $E^\circ$ will act as the Oxidizing Agent (Cathode) and force the species with the lower $E^\circ$ to undergo oxidation (Anode).
Applications of Electrochemical Series
- Calculating Standard Cell Potential ($E^\circ_{cell}$):
$E^\circ_{cell} = E^\circ_{cathode} - E^\circ_{anode}$ (Using reduction potentials for both).
- Predicting Reaction Feasibility: For a redox reaction to be spontaneous, the $E^\circ_{cell}$ must be positive. This means the Gibbs Free Energy ($\Delta G^\circ = -nFE^\circ_{cell}$) is negative.
- Displacing Hydrogen from Acids: Only metals with a negative standard reduction potential (placed below Hydrogen in the ECS) can displace $H_2$ gas from dilute acids. Copper ($E^\circ = +0.34\text{V}$) cannot react with dilute HCl!