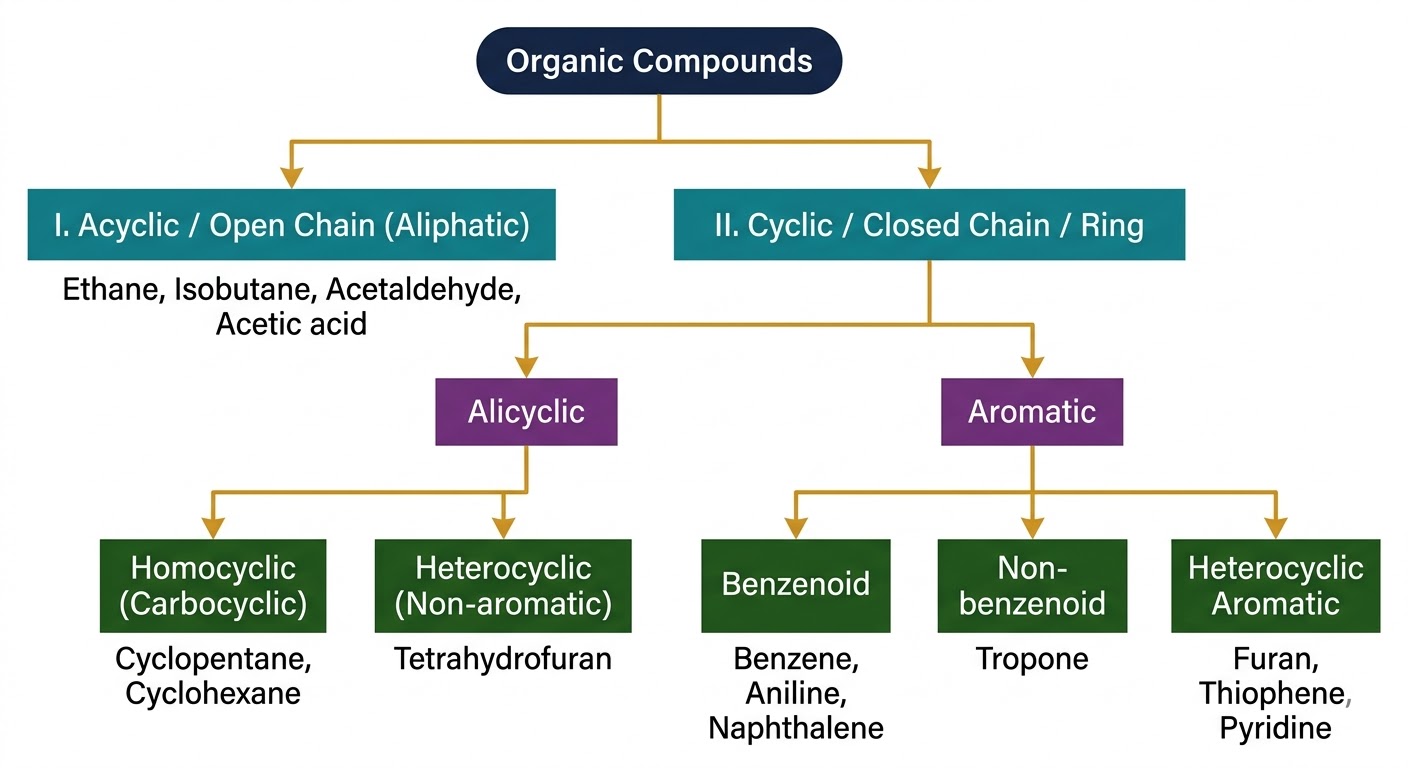
Organic compounds are vital for sustaining life on Earth — from the DNA that carries our genetic code to the proteins in our blood, muscles and skin. They also appear in fuels, polymers, dyes, medicines, and clothing.
Organic Chemistry is the branch of chemistry dealing with carbon compounds — primarily those formed with H, O, N, S, P, and halogens through covalent bonds.
Catenation: Carbon's unique ability to form covalent bonds with other carbon atoms — the root of organic chemistry's diversity.
| Year | Scientist | Event |
|---|---|---|
| ~1780 | Berzilius (Sweden) | Proposed Vital Force Theory — organic compounds can only be made inside living organisms by a mysterious "vital force" |
| 1828 | F. Wöhler | Disproved Vital Force — synthesised urea (organic) from ammonium cyanate (inorganic) in a lab |
| 1845 | Kolbe | Synthesised acetic acid from inorganic sources |
| 1856 | Berthelot | Synthesised methane from inorganic sources |
| Later | — | Electronic theory of covalent bonding → modern organic chemistry |
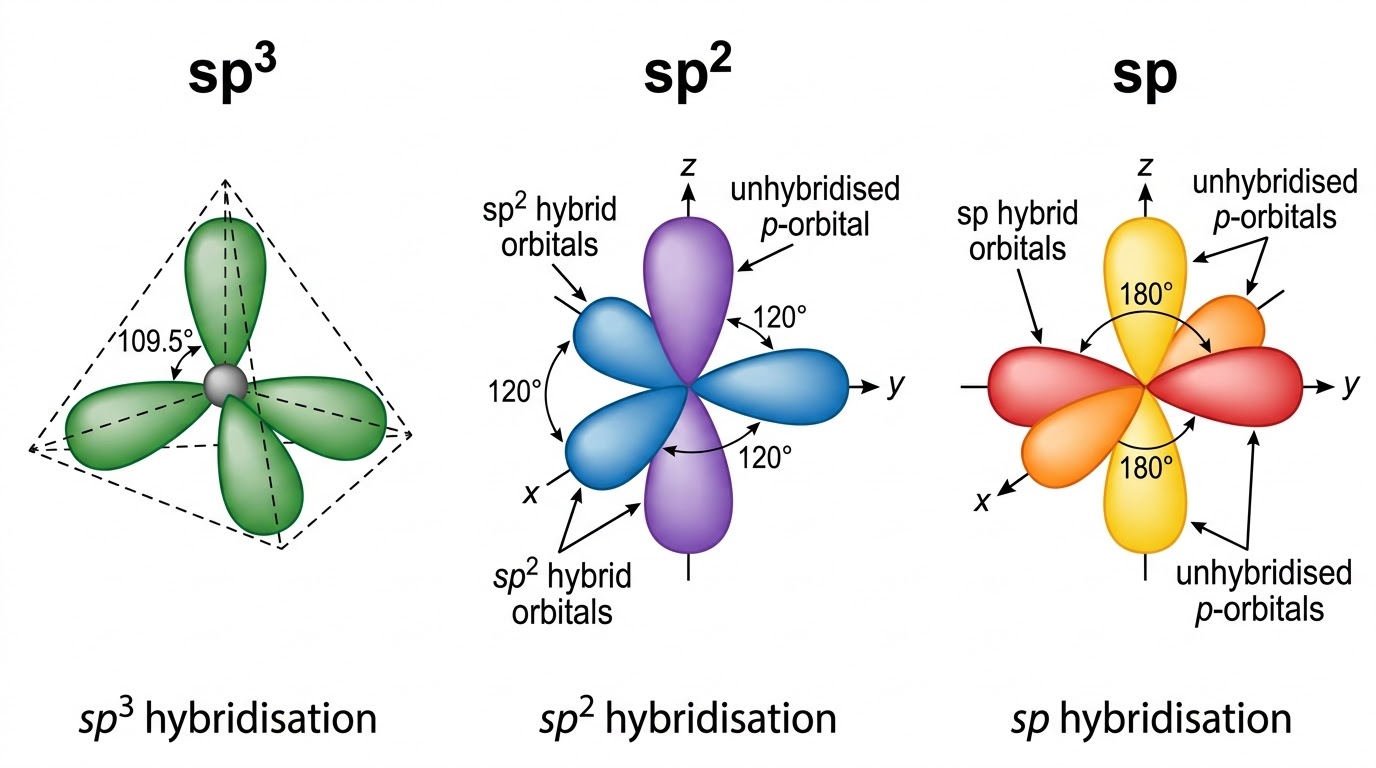
Carbon (atomic number 6, config 1s²2s²2p²) has 4 valence electrons and always forms 4 bonds. The shape depends on hybridisation.
| Hybridisation | s character | Shape | Bond Angle | Example | Bond character |
|---|---|---|---|---|---|
| sp³ | 25% | Tetrahedral | 109.5° | CH4 | Longest, weakest, least electronegative C |
| sp² | 33% | Trigonal Planar | 120° | C2H4 | Intermediate length & strength |
| sp | 50% | Linear | 180° | C2H2 | Shortest, strongest; most electronegative C |
Greater s-character → Greater electronegativity of C → Shorter & stronger bonds formed.
Electronegativity order: sp (50% s) > sp² (33% s) > sp³ (25% s)
.jpg)
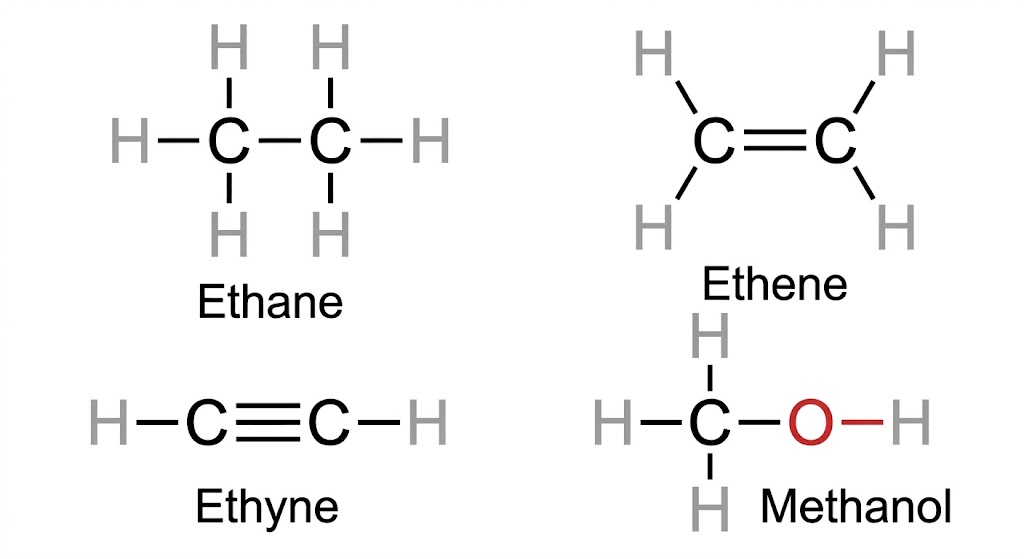
| Type | Description | Example – Ethane |
|---|---|---|
| Complete / Lewis Structure | Every bond shown as a dash; all atoms written; lone pairs may be shown | H–C(H)(H)–C(H)(H)–H (full expanded) |
| Condensed Formula | Dashes omitted; identical groups shown with subscripts | CH3CH3 |
| Bond-line (Skeletal) Formula | C and H not shown; C–C bonds as zig-zag lines; heteroatoms written | A straight zig-zag line (2 carbons) |
Aromatic compounds are centred around the benzene ring. All substituents are shown in matching amber style, properly connected to the ring.
An atom or group of atoms joined to the carbon chain that is responsible for the characteristic chemical properties of the compound.
Examples: –OH (hydroxyl), –CHO (aldehyde), –COOH (carboxylic acid), –NH2 (amino), C=C (alkene), C≡C (alkyne)
A series of organic compounds each containing the same functional group; each successive member differs from the previous by –CH2– (14 mass units). Members are called homologues.
IUPAC = International Union of Pure and Applied Chemistry. In this system, names are correlated with structure so that the reader can deduce structure from name.
Suffix: –ane. Prefix = number of carbons.
| Name | Formula | Name | Formula |
|---|---|---|---|
| Methane | CH4 | Heptane | C7H16 |
| Ethane | C2H6 | Octane | C8H18 |
| Propane | C3H8 | Nonane | C9H20 |
| Butane | C4H10 | Decane | C10H22 |
| Pentane | C5H12 | Icosane | C20H42 |
| Hexane | C6H14 | Triacontane | C30H62 |
Always prefix (never suffix): –R, C6H5– (phenyl), –F, –Cl, –Br, –I, –NO2, alkoxy (–OR)
Isomerism: Two or more compounds with the same molecular formula but different properties. Such compounds are isomers.
| Type | Cause | Example |
|---|---|---|
| Chain Isomerism | Different carbon skeletons | C5H12: Pentane, 2-Methylbutane (Isopentane), 2,2-Dimethylpropane (Neopentane) |
| Position Isomerism | Same skeleton & functional group, different position | C3H8O: Propan-1-ol vs Propan-2-ol |
| Functional Group Isomerism | Same formula, different functional groups | C3H6O: Propanone (ketone) vs Propanal (aldehyde) |
| Metamerism | Different alkyl chains on either side of the same functional group | C4H10O: CH3OC3H7 vs C2H5OC2H5 |
| Stereoisomerism – Geometrical | Same bonds, different spatial arrangement due to restricted rotation (C=C) | cis-But-2-ene vs trans-But-2-ene |
| Stereoisomerism – Optical | Same bonds, non-superimposable mirror images (chiral centre) | L- and D-lactic acid |
.jpg)
Shared pair stays with one fragment → charged species.
Each fragment gets one electron → neutral radicals.
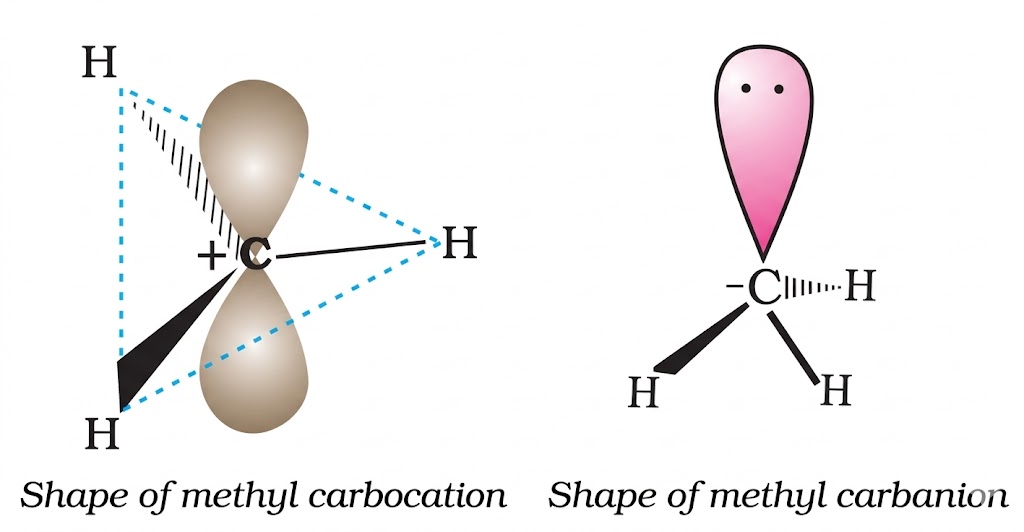
Reason: More alkyl groups → more hyperconjugation + inductive stabilisation → more stable carbocation.
= Nucleus-seeker. Electron-rich. Donates electron pair to electrophile.
Charged: HO–, CN–, HS–, C2H5O–, R3C:–
Neutral with lone pair: H2O, NH3, R3N, ROH
= Electron-seeker. Electron-deficient. Accepts electron pair from nucleophile.
Positive ions: H+, NO2+, CH3+
Neutral: BF3, AlCl3, SO3; R–X (δ+ on C), >C=O
.jpg)
| Effect | Nature | Transmission | Key Feature |
|---|---|---|---|
| Inductive | Permanent, ground state | Through σ-bonds; decreases rapidly; almost zero after 3 bonds | Electron withdrawal (–I: halogens, –NO2, –CN) or donation (+I: alkyl groups) |
| Resonance (Mesomeric) | Permanent, ground state | Through conjugated π system | +R (electron away from group): –OH, –NH2, halogens | –R (electrons toward group): –COOH, –CHO, –NO2, –CN |
| Electromeric | Temporary — only when reagent approaches | Complete π electron transfer to one atom on demand | +E: electrons go to atom where reagent attaches | –E: electrons go away from attachment point. Annulled when reagent removed. |
| Hyperconjugation | Permanent | σ(C–H) electrons → adjacent π or empty p orbital | "No-bond resonance". More alkyl groups = more stabilisation of carbocation/free radical/alkene |
Benzene's actual C–C bond length = 139 pm — intermediate between C–C single (154 pm) and C=C double (134 pm). This proves it is a resonance hybrid, not alternating single/double bonds.
.jpg)
Hyperconjugation = σ(C–H) electrons of an adjacent alkyl group delocalise into the empty p-orbital of a carbocation or π-system. This disperses the positive charge and stabilises the species.
| Method | Principle | Used For | Key Details |
|---|---|---|---|
| Sublimation | Solid → vapour directly (bypasses liquid state) | Sublimable compounds from non-sublimable impurities | Camphor, iodine, naphthalene, benzoic acid |
| Crystallisation | Difference in solubility at different temperatures | Purification of solid organic compounds | Dissolve at high T in minimum solvent → cool → filter crystals. Activated charcoal removes coloured impurities. |
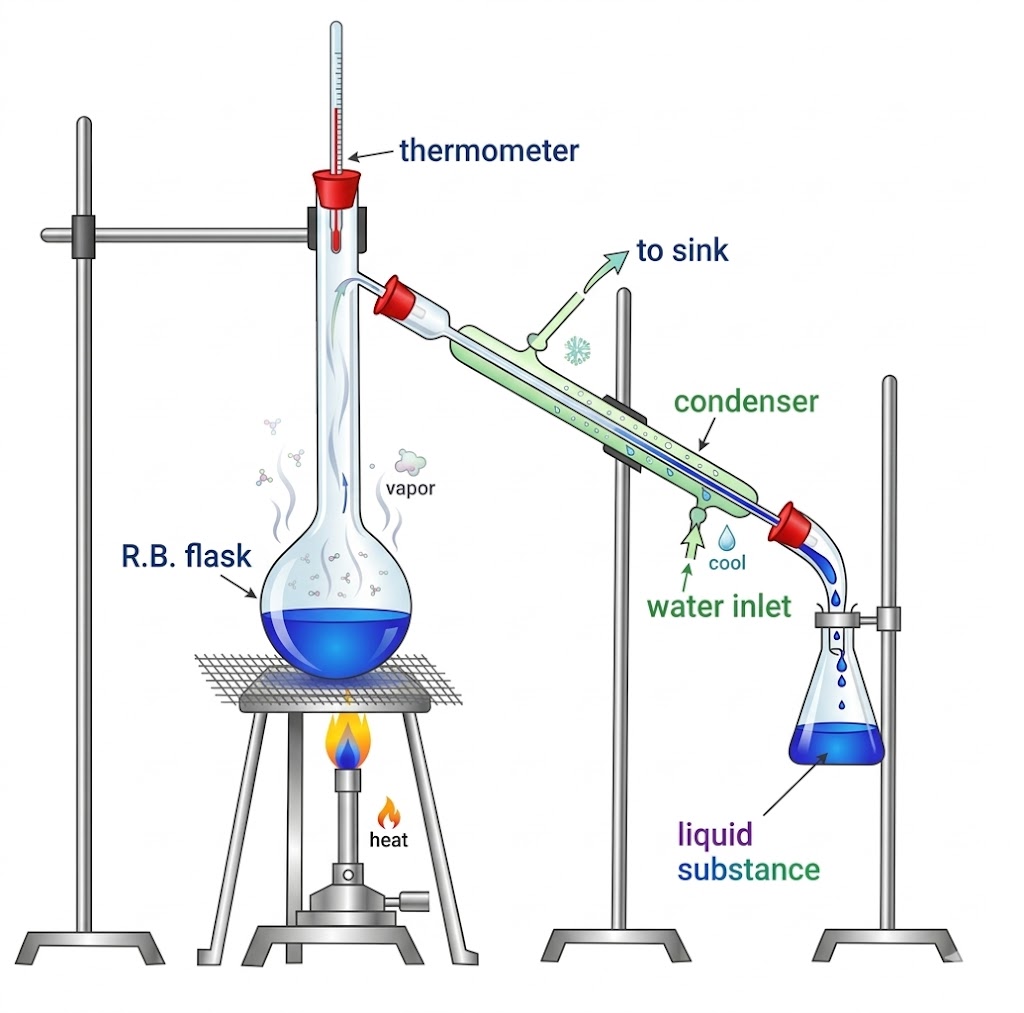
| Simple Distillation | Difference in boiling points | Volatile liquid from non-volatile impurities; liquids with large bp difference | CHCl3 (bp 334 K) from aniline (bp 457 K) |
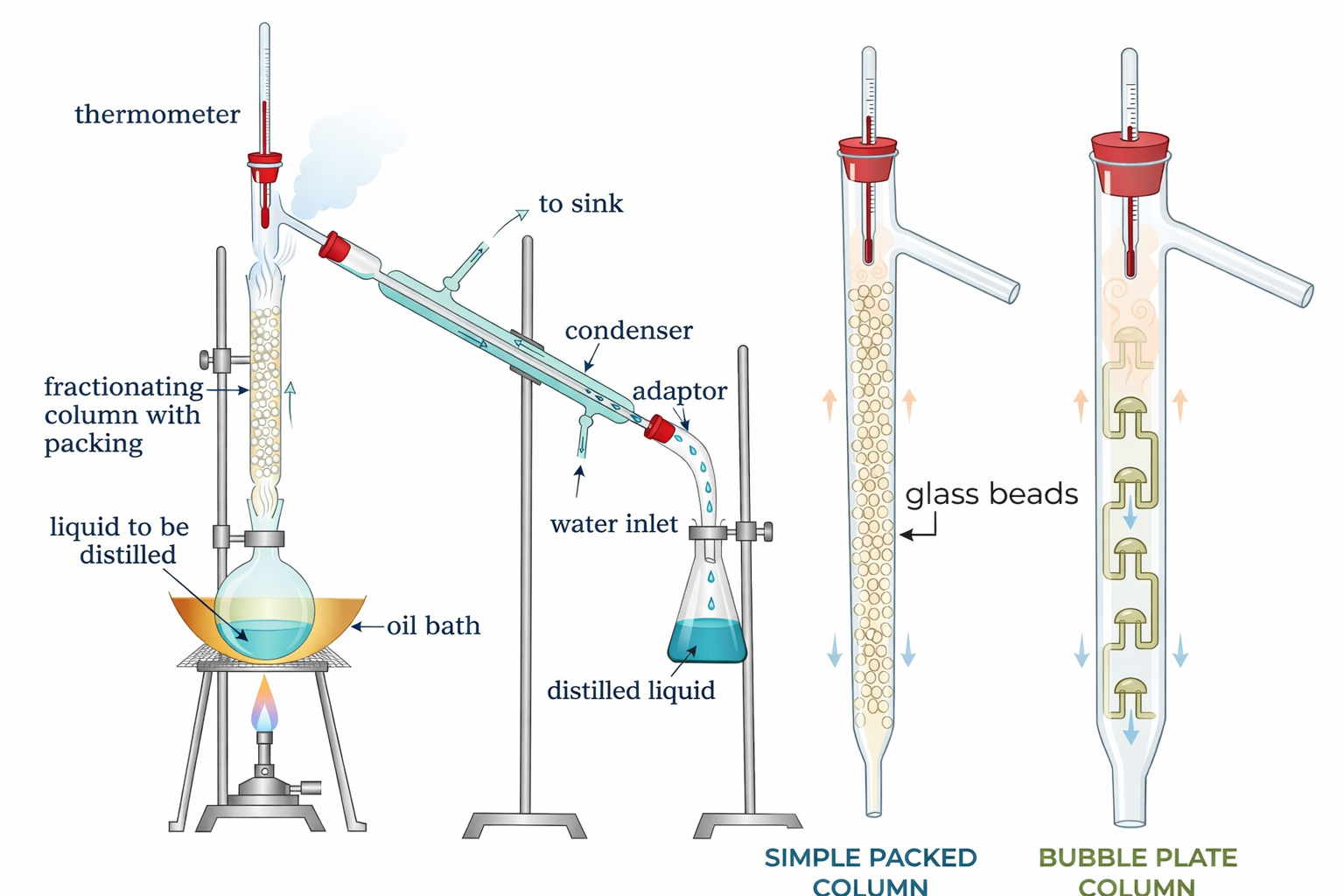
| Fractional Distillation | Same + fractionating column (multiple condensation-vaporisation steps) | Liquids with close boiling points (e.g. petroleum) | Each step = "theoretical plate". Commercially: hundreds of plates. |
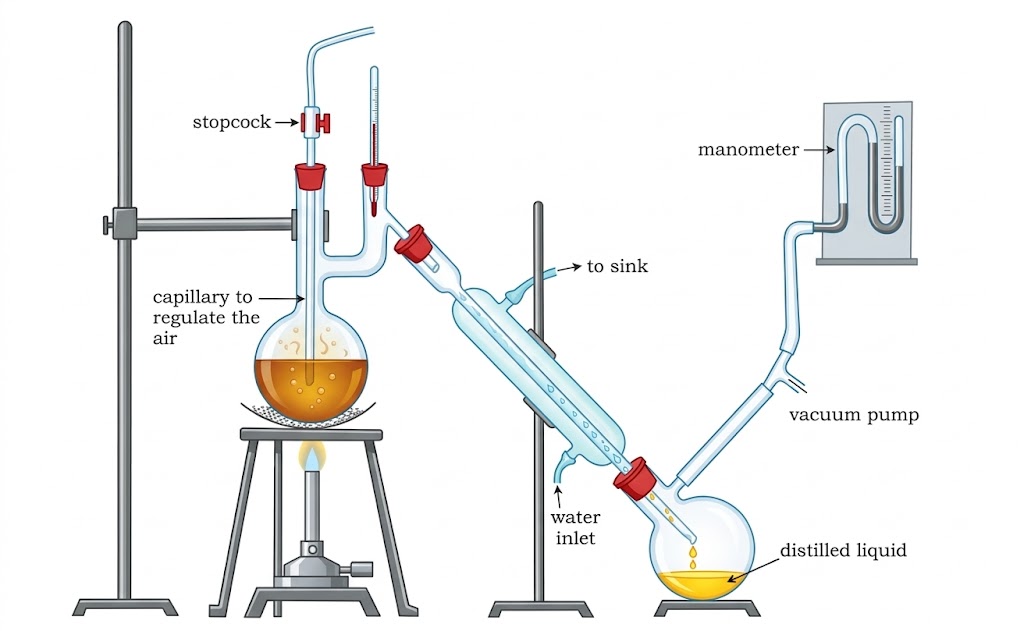
| Vacuum Distillation | Liquid boils when vapour pressure = reduced external pressure | Liquids with very high bp or those that decompose at their bp | Glycerol in soap industry. Uses water pump / vacuum pump. |
| Steam Distillation | p(total) = p₁(organic) + p₂(water); boiling below 373 K | Steam volatile, water-immiscible compounds | Aniline separation. Since p₁ < p (atm), boiling occurs below 373 K. |
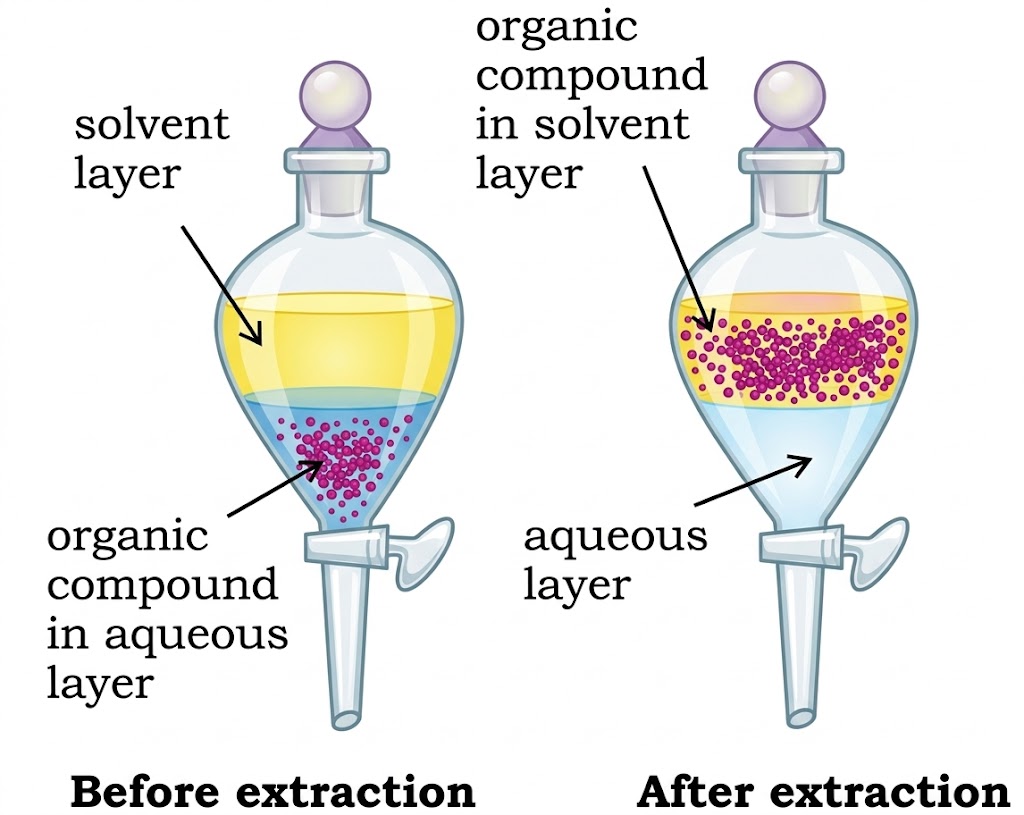
| Differential Extraction | Organic compound more soluble in organic solvent than water | Extracting organic compound from aqueous medium | Separating funnel; two immiscible layers form; organic layer separated. |
| Chromatography | Differential migration between stationary and mobile phases | Separation, purification, identification | Best modern technique. TLC, column, paper chromatography. |
Rf is characteristic of a compound in a given solvent system. Range: 0 to 1. Different compounds have different Rf values → basis of separation and identification.
 Developed chromatogram sh.jpg)
Heat compound with copper(II) oxide (CuO). Carbon → CO2 (tested with lime water); Hydrogen → H2O (tested with anhydrous CuSO4).
Principle: Organic compound fused with metallic sodium → converts covalent N, S, X into ionic (water-soluble) forms: NaCN, Na2S, NaX. The fused mass dissolved in distilled water = Sodium Fusion Extract (SFE).
| Element | Reagent Added to SFE | Positive Test Observation | Key Reaction |
|---|---|---|---|
| Nitrogen | Boil with FeSO4 → acidify with conc. H2SO4 | Prussian blue colour | 6CN– + Fe2+ → [Fe(CN)6]4– then 3[Fe(CN)6]4– + 4Fe3+ → Fe4[Fe(CN)6]3 (Prussian blue) |
| Sulphur (a) | Acidify with acetic acid + lead acetate | Black precipitate (PbS) | S2– + Pb2+ → PbS↓ (black) |
| Sulphur (b) | Sodium nitroprusside solution | Violet colour | S2– + [Fe(CN)5NO]2– → [Fe(CN)5NOS]4– (violet) |
| Chlorine | Acidify with HNO3 + AgNO3 | White ppt — soluble in NH4OH | Cl– + Ag+ → AgCl↓ (white) |
| Bromine | Acidify with HNO3 + AgNO3 | Pale yellow ppt — sparingly soluble in NH4OH | Br– + Ag+ → AgBr↓ (pale yellow) |
| Iodine | Acidify with HNO3 + AgNO3 | Yellow ppt — insoluble in NH4OH | I– + Ag+ → AgI↓ (yellow) |
| Phosphorus | Oxidise with Na2O2 → boil with HNO3 → ammonium molybdate | Yellow ppt (ammonium phosphomolybdate) | H3PO4 + 12(NH4)2MoO4 + 21HNO3 → (NH4)3PO4·12MoO3↓ |
N + S both present: NaSCN forms → gives blood red colour with Fe3+ (no Prussian blue since no free CN–). With excess Na: NaSCN decomposes → NaCN + Na2S → normal tests work.
Before halogen test: If N or S also present → first boil SFE with conc. HNO3 (to destroy NaCN and Na2S which would precipitate as AgCN/Ag2S and give false positives).
Why acetic acid, not H2SO4 in sulphur test? H2SO4 provides SO42– → PbSO4 precipitate (false positive). Acetic acid = inert, no interference.
Known mass of compound burned in excess O2 with CuO. CO2 absorbed in KOH U-tube; H2O absorbed in CaCl2 U-tube. Increase in mass of each U-tube gives the amounts.
0.246 g of organic compound gave 0.198 g CO2 and 0.1014 g H2O on combustion.
% C = (12 × 0.198 × 100) ÷ (44 × 0.246) = 21.95%
% H = (2 × 0.1014 × 100) ÷ (18 × 0.246) = 4.58%
.jpg)
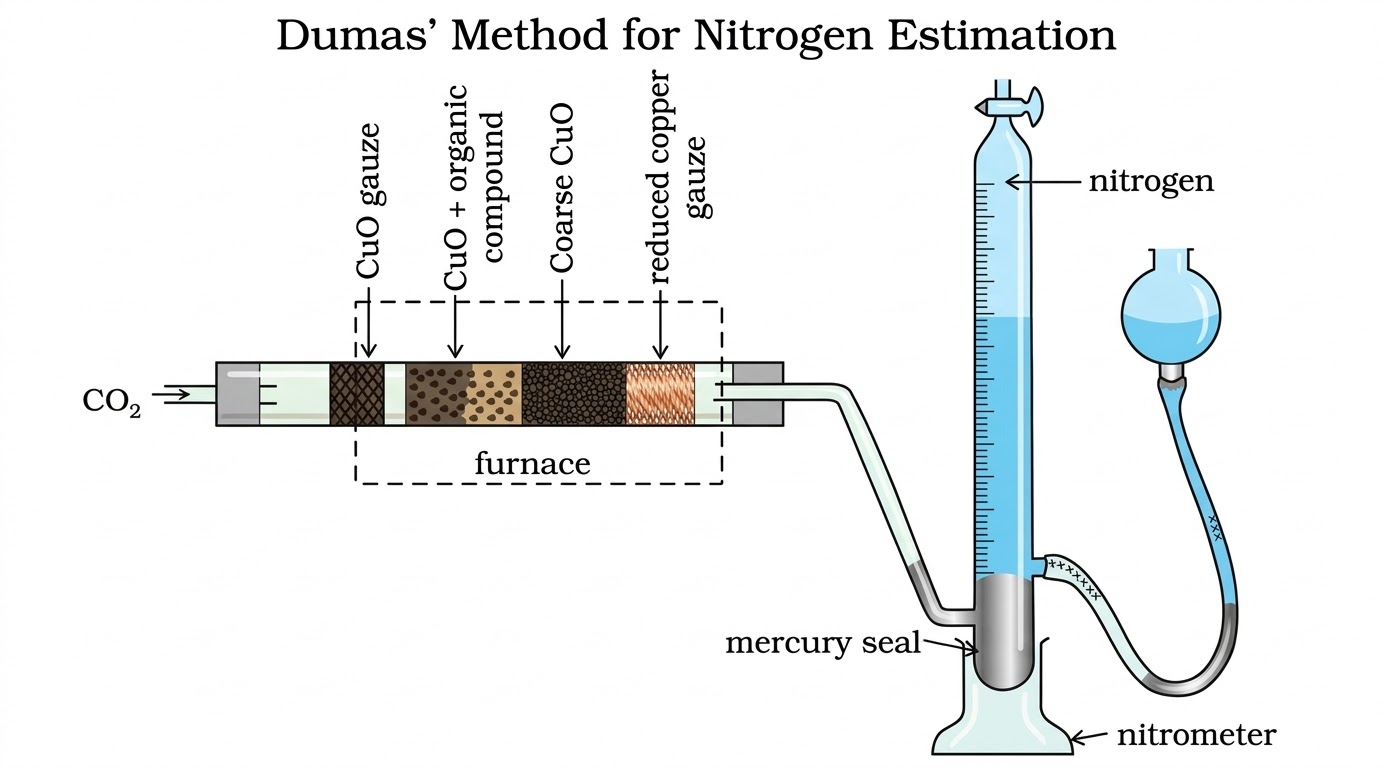
Compound + CuO in CO2 atmosphere → N2 gas collected over KOH. Measure volume.
Convert volume to STP: $V_{STP} = \dfrac{p_1 V_1 \times 273}{760 \times T_1}$
where p1 = atmospheric pressure − aqueous tension
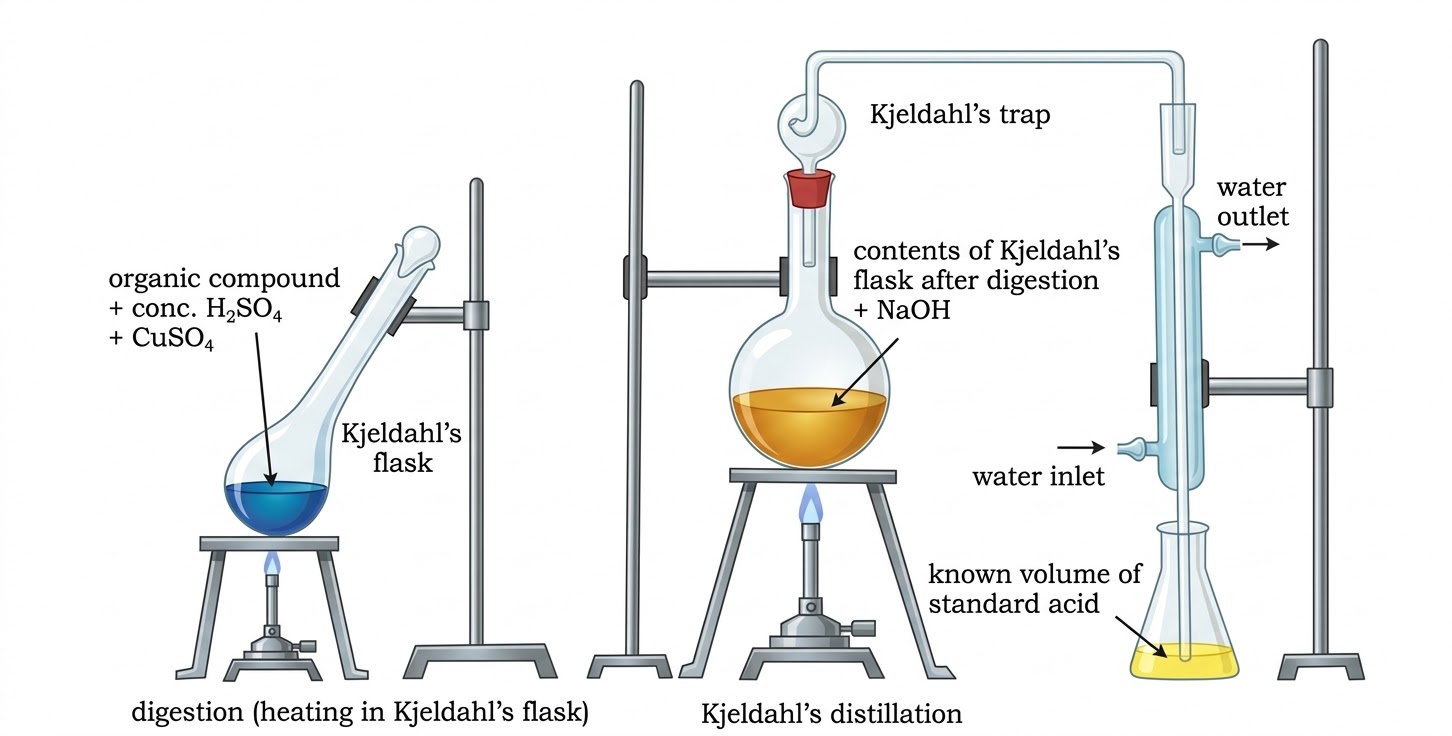
$$\% N = \frac{28 \times V_{STP} \times 100}{22400 \times m}$$Compound + conc. H2SO4 → (NH4)2SO4; then + excess NaOH → NH3 gas absorbed in standard H2SO4; back-titrate unreacted acid with NaOH.
$$\% N = \frac{1.4 \times M \times 2(V - V_1/2)}{m}$$M = molarity of H2SO4, V = vol of H2SO4 taken, V1 = vol of NaOH used, m = mass of compound.
NOT applicable to: –NO2 (nitro), azo groups, N in ring (e.g. pyridine).
0.3 g compound → 50 mL N2 at 300 K and 715 mm pressure. Aqueous tension at 300 K = 15 mm.
Actual pressure = 715 – 15 = 700 mm
Volume at STP = (273 × 700 × 50) ÷ (300 × 760) = 41.9 mL
% N = (28 × 41.9 × 100) ÷ (22400 × 0.3) = 17.46%
Compound + fuming HNO3 + AgNO3 → sealed Carius tube in furnace → halogen forms silver halide (AgX). Filter, dry, weigh AgX.
0.15 g compound → 0.12 g AgBr. M(AgBr) = 108 + 80 = 188 g/mol
% Br = (80 × 0.12 × 100) ÷ (188 × 0.15) = 34.04%

| Element | Method | Product Measured | Formula |
|---|---|---|---|
| C | Liebig (CuO combustion) | CO2 absorbed in KOH U-tube | % C = 12×m2×100 / (44×m) |
| H | Liebig (CuO combustion) | H2O absorbed in CaCl2 U-tube | % H = 2×m1×100 / (18×m) |
| N | Dumas / Kjeldahl's | N2 gas / NH3 absorbed in H2SO4 | See formulas above |
| X (halogens) | Carius (fuming HNO3 + AgNO3) | AgX (silver halide) | % X = at.mass(X)×m1×100 / (M(AgX)×m) |
| S | Carius (fuming HNO3) | BaSO4 precipitate | % S = 32×m1×100 / (233×m) |
| P | Fuming HNO3 + ammonium molybdate | (NH4)3PO4·12MoO3 | % P = 31×m1×100 / (1877×m) |
| O | By difference | — | % O = 100 − Σ(all other %) |