
CBSE Class 11 Physics • Chapter 11
Chapter Overview
Welcome to Thermodynamics, a central pillar of Physics! This chapter deals with concepts of heat, temperature, and the rigorous mathematical interconversion of heat and other forms of energy. We will deeply analyze the macroscopic properties of matter (like Pressure, Volume, and Temperature) and how they dictate the rules of engines, refrigerators, and the universe itself.
Thermodynamics is the branch of physics that deals with the concepts of heat and temperature and the inter-conversion of heat and other forms of energy. It is a macroscopic science. This means we do not care about the individual motion of trillions of molecules (like in Kinetic Theory); instead, we care about bulk properties that we can actually measure with instruments.
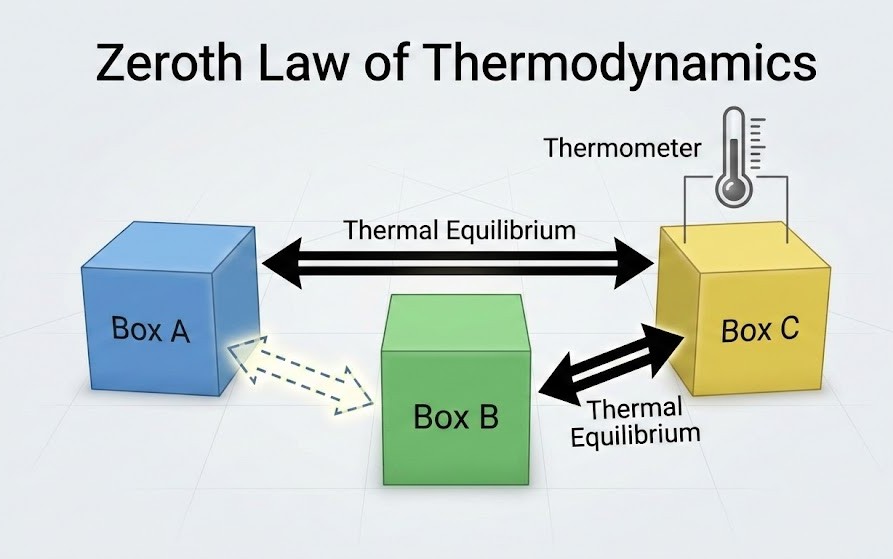
What is Thermal Equilibrium?
A system is in thermal equilibrium if its macroscopic variables ($P, V, T, \text{mass}$) do not change with time. If you put two systems in contact via a conducting (diathermic) wall, they will exchange heat until their states stop changing. When the heat flow stops, they are said to be in Thermal Equilibrium.
Zeroth Law of Thermodynamics (NCERT Statement):
"If two systems A and B are each in thermal equilibrium with a third system C, then A and B are in thermal equilibrium with each other."
Why is this important?
It gives us the formal definition of Temperature. The Zeroth Law implies that all
bodies in thermal equilibrium must have a common physical property. We call this property Temperature!
Thus, temperature is the scalar quantity that determines whether or not two bodies are in thermal
equilibrium.
1. Heat ($\Delta Q$): It is the energy that is transferred between a system and its environment because of a temperature difference between them. Heat is energy in transit. It is not stored in a body.
2. Work ($\Delta W$): Work is the energy transferred by the system to its surroundings via macroscopic mechanical forces (like a gas pushing a piston). Work is also energy in transit!
3. Internal Energy ($U$): This is the total intrinsic energy stored inside the
system. It is the sum of the kinetic and potential energies of all its molecules.
Crucially, for an Ideal Gas, there is no intermolecular potential energy, so its
Internal Energy is purely kinetic and depends only on its absolute Temperature ($U \propto
T$).
Path Function vs State Function (NCERT Clarification)
The First Law is simply the principle of Conservation of Energy applied to a thermodynamic system.
Statement: The total amount of heat energy supplied to a system ($\Delta Q$) is equal to the sum of the increase in the internal energy of the system ($\Delta U$) and the external work done by the system ($\Delta W$).
In differential calculus form (for infinitesimally small changes):
Crucial Sign Convention (Physics):
(Note: In Chemistry, the IUPAC convention for Work is often reversed. Be careful! In Physics, we care about the work the gas pushes out.)
Molar Specific Heat ($C$): The amount of heat required to raise the temperature of 1 mole of a gas by $1^\circ C$ (or 1 K). $\Delta Q = \mu C \Delta T$. Because gases can expand, the heat required depends on the conditions of heating.
Derivation of Mayer's Relation ($C_p - C_v = R$) from NCERT:
Let's consider $\mu = 1$ mole of an ideal gas. According to the First Law ($dQ = dU + dW$) where $dW = P dV$.
Case 1: Heating at Constant Volume (Isochoric)
Since Volume is constant, $dV = 0 \implies dW = 0$.
So, $dQ = dU$. By definition, heat supplied at constant volume is $dQ = C_v dT$.
Therefore, $dU = C_v dT \implies C_v = \frac{dU}{dT}$. (This establishes that internal energy solely depends on $C_v$ and temperature!).
Case 2: Heating at Constant Pressure (Isobaric)
Let the same gas be heated by the same $dT$ but at constant Pressure $P$. It expands by $dV$.
Heat supplied: $dQ = C_p dT$.
Work done: $dW = P dV$. From ideal gas law $PV = RT$, at constant $P$, $P dV = R dT$. Therefore, $dW = R dT$.
Using First Law: $dQ = dU + dW$
Substitute our findings: $C_p dT = C_v dT + R dT$
Dividing throughout by $dT$ yields Mayer's Formula:
Every equilibrium state of a system is completely described by specific variables.
The mathematical relationship connecting these state variables is called the Equation of State. For an ideal gas, it is:
Where $\mu$ is the number of moles, and $R$ is the universal gas constant ($8.314 \text{ J mol}^{-1} \text{ K}^{-1}$).
A thermodynamic process happens when a system undergoes a change from one equilibrium state $(P_1, V_1, T_1)$ to another $(P_2, V_2, T_2)$.
Figure 11.1: The Four Key Thermodynamic Processes on a P-V Indicator Diagram
Condition: Temperature remains strictly constant ($dT = 0$). To achieve this, the cylinder walls must be perfectly conducting, and the process must occur very slowly so heat can exchange with the surroundings to keep temperature stable.
Equation of state: From $PV = \mu RT$, since $T$ is constant, we get Boyle's Law: $$ PV = \text{Constant} $$
Work Done Derivation ($W$):
$$ W = \int_{V_1}^{V_2} P \, dV $$
Substitute $P = \frac{\mu RT}{V}$:
$$ W = \int_{V_1}^{V_2} \frac{\mu RT}{V} \, dV = \mu RT \int_{V_1}^{V_2} \frac{1}{V} \, dV $$
Since the integral of $1/V$ is $\ln V$, we get:
First Law Check: Since $dT = 0 \implies \Delta U = 0$. Therefore, $dQ = dW$. All heat absorbed by the gas is entirely used to do external work!
Condition: No heat enters or leaves the system ($dQ = 0$). To achieve this, the cylinder must be perfectly insulated (adiabatic walls), or the process must occur so fast that there isn't time for heat transfer (like a bursting tire).
Equation of state: The relationship follows Poisson's Law:
(Where $\gamma = C_p / C_v$ is the ratio of specific heats).
Work Done Derivation ($W$):
Let $PV^\gamma = K$. Then $P = \frac{K}{V^\gamma}$.
$$ W = \int_{V_1}^{V_2} \frac{K}{V^\gamma} \, dV = K \int_{V_1}^{V_2} V^{-\gamma} \, dV $$
Using the power rule for integration $\int x^n dx = \frac{x^{n+1}}{n+1}$:
$$ W = K \left[ \frac{V^{1-\gamma}}{1-\gamma} \right]_{V_1}^{V_2} = \frac{K V_2^{1-\gamma} - K V_1^{1-\gamma}}{1-\gamma} $$
Substitute $K = P_2 V_2^\gamma$ in the first term, and $K = P_1 V_1^\gamma$ in the second term:
First Law Check: Since $dQ = 0$, the first law gives $0 = \Delta U + \Delta W \implies \Delta W = -\Delta U$. If the gas expands (positive work), it does so at the expense of its own internal energy, which is why an adiabatic expansion causes dramatic cooling!
What is a Heat Engine? It's a device that takes heat from a hot source, converts a portion of it into useful mechanical work, and dumps the leftover waste heat into a cold sink. This operates in a repeating continuous cycle.
Main Components:
Working Diagram & Efficiency ($\eta$):
The engine absorbs heat $Q_1$ from the Source, does useful Work $W$, and rejects remaining heat $Q_2$ to the Sink.
By energy conservation: $Q_1 = W + Q_2 \implies W = Q_1 - Q_2$
Efficiency Formula: Efficiency is defined as Output / Input.
Because some heat $Q_2$ must ALWAYS be rejected, $\eta$ can never be 1 (100%).
A refrigerator is exactly a heat engine running in reverse! It takes heat from a cold body (the freezer), external Work ($W$) is done on the compressor by electricity, and it dumps a larger total amount of heat into the hot body (the kitchen).
Coefficient of Performance ($\alpha$ or $\beta$): Instead of efficiency, we measure how much cooling it did for the electricity we paid for.
Unlike efficiency, $\alpha$ can easily be greater than 1.
The First Law says energy is conserved. But it does not tell you the direction of heat transfer. Can we take heat from a cold rock and make an engine work? The First Law doesn't forbid it. The Second Law establishes the fundamental asymmetry of nature.
1. Kelvin-Planck Statement (Engine Rule):
"No process is possible whose sole result is the absorption of heat from a reservoir and the complete
conversion of the heat into work."
Basically: You can't make an engine that is 100% efficient. You MUST have a cold sink to dump waste
heat.
2. Clausius Statement (Refrigerator Rule):
"No process is possible whose sole result is the transfer of heat from a colder object to a hotter
object."
Basically: Heat won't flow from cold to hot naturally. You must plug in a compressor (do external work)
to force it to happen.
NCERT Note: These two statements look different but are completely mathematically equivalent!
Sadi Carnot, a brilliant French engineer, proposed a theoretical, idealized, completely reversible heat engine operating between two temperatures $T_1$ and $T_2$. He proved mathematically that no real engine operating between two temperatures can have a greater efficiency than a Carnot engine operating between the same two temperatures.
Figure 11.3: Carnot Cycle on a P-V Indicator Diagram
The Four Strokes of the Carnot Cycle:
Notice that the net work from the adiabatic steps cancels out ($W_2 = -W_4$).
After complex but elegant mathematical ratio simplifications (using the adiabatic relation $T V^{\gamma-1} = const$ to prove that $V_2/V_1 = V_3/V_4$), the heat ratio perfectly mirrors the absolute temperature ratio:
$$ \frac{Q_2}{Q_1} = \frac{T_2}{T_1} $$
Therefore, the Efficiency of an Ideal Carnot Engine is:
Carnot Theorem: This elegant formula $\eta = 1 - T_2/T_1$ proves that efficiency depends
ONLY on the temperatures of the Source and Sink, and is completely independent of the nature of the
working substance (whether it's steam, perfect gas, or anything else).
To get 100% efficiency, the
sink must be at Absolute Zero ($0 \text{ K}$), which is practically impossible!
| Concept | Formula | Significance |
|---|---|---|
| First Law of Thermo | $\Delta Q = \Delta U + \Delta W$ | Energy conservation macroscopically. |
| Internal Energy (Ideal Gas) | $U = \frac{f}{2} \mu R T$ | Depends purely on Temperature (state variable). |
| Mayer's Formula | $C_p - C_v = R$ | Connects specific heats due to expansion work. |
| Isothermal Work | $W = \mu R T \ln\left(\frac{V_2}{V_1}\right)$ | Work done while keeping $T$ constant. |
| Adiabatic Equation of State | $P V^\gamma = \text{Constant}$ | Relation with no heat exchange. |
| Adiabatic Work | $W = \frac{\mu R(T_1 - T_2)}{\gamma - 1}$ | Work done using only internal energy. |
| Engine Efficiency | $\eta = 1 - \frac{Q_2}{Q_1}$ | Output Work / Input Heat. |
| Carnot Efficiency | $\eta = 1 - \frac{T_2}{T_1}$ | Absolute maximum possible efficiency for any engine given $T_1$, $T_2$. |